Abstract
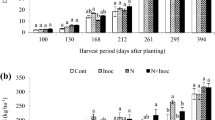
TheA-value method, involving the application of a higher15N rate to a reference non-N2-fixing plant, was used to assess the magnitude of N2 fixation in two bambara groundnut cultivars at four growth stages [vegetative, 0–47 days after planting (DAP); early pod-filling, 47–99 DAP; mid-pod-filling, 99–120 DAP; physiological maturity, 120–148 DAP). The cultivars were Ex-Ada, a bunchy type, and CS-88-11, a slightly spreading type. They were grown on a loamy sand. Uninoculated Ex-Ada and CS-88-11 were used as reference plants to measure the N2 fixed in the inoculated bambara groundnuts. In this greenhouse study, soil was the major source of N in bambara groundnuts during vegetative growth, and during this period it accounted for over 80% of the N accumulaed in the plants. However, N2 fixation became the major source of plant N during reproductive growth. There were significant differences between the two cultivars in the ability to fix N2, and at physiological maturity, almost 75% of the N in CS-88-11 was derived from the atmosphere compared to 55% in Ex-Ada. Also, the total N fixed in CS-88-11 at physiological maturity was almost double that in Ex-Ada. Our data indicate that the higher N2 fixation in CS-88-11 was due to two factors, a higher intensity of N2 fixation and a longer active period of N2 fixation. The results also suggest that bambara groundnut genotypes could be selected for higher N2 fixation in farining systems.
Similar content being viewed by others
References
Dakora FD, Atkins CA, Pate JS (1992) Effect of NO sup-inf3 of N2 fixation and nitrogenous solutes of xylem in two nodulated West African Geocarpic legumes, Kersting's bean (Macrotyloma geocarpum L.) and bambara groundnut (Vigna unguiculata L.). Plant and Soil 140:225–262
Danso SKA (1986) Methods for estimating biological nitrogen fixation. In: Ssali H, Keya SO (eds) Biological nitrogen fixation in Africa. Mircen, Nairobi, pp, 224–244
Danso SKA (1985) Review: Estimation of N2-fixation by isotope dilution: An appraisal of techniques involving15N enrichment and their application—Comments. Soil Biol Biochem 18:243–244
Danso SKA, Alexander M (1974) Survival of two strains ofRhizobium in soil. Soil Sci Soc Am Proc 38:86–89
Danso SKA, Hera C, Douka C (1987) Nitrogen fixation in soybean as influenced by cultivar andRhizobium strain. Plant and Soil 99:163–174
Doku EV, Karikari SK (1971) Bambara groundnut. Econ Bot 25:255–262
Duke JA, Okigbo DN, Reed CF, Weder JKP (1977)Voandzeia subterranea (L) Thouars. Trop Grain Legume Bull 10:8–11
Fiedler R, Proksch G (1975) The determination of15N by emission and mass spectrometry in biochemical analysis. Ann Chim Acta 78:1–62
Fried M, Danso SKA, Zapata F (1983) The methodology of measurement of N2 fixation by non-legumes as inferred from field experiments with legumes. Can J Microbiol 29:1053–1062
Gueye M, Bordeleau LM (1988) Nitrogen fixation in bambara groundnut (Voandzeia subterranea (L) Thouars. Mircen J 4:365–375
Hardarson G, Zapata F, Danso SKA (1984) Effect of plant genotype on symbiotic nitrogen fixation by soybean cultivars Plant and Soil 82:397–405
Hardarson G, Danso SKA, Zapata F, Reichardt K (1991) Measurement of nitrogen fixation at different N-fertilizer rates using15N isotope dilution and “A” value methods. Plant and Soil 131: 161–168
Henson RA, Heichel GH (1984) Partitioning of symbiotically fixed nitrogen in soybeans and alfalfa. Crop Sci 24:986–990
Hoagland DR, Aron DI (1938) The water culture method for growing plants without soil. Calif Agric Exp Sta Circ no. 347
Kaleem F (1988) Nitrogen fixation by monocropped and intercropped legumes and their residual effect on a following maize crop. In: Nyankpala Agric Exp Sta (Crops Res Inst) Annu Rep, pp 176–181
Kumarasinghe KS, Danso SKA, Zapata F (1992) Field evaluation of N2 fixation and N partitioning in climbing bean (Phaseolus vulgaris L.) using15N. Biol Fertill Soils 13:142–146
Mulongoy K, Goli AE (1988) Nodulation and growth of bambara groundnut (Vigna subterranea) in three soils. In: Maximiser La Fixation Biologique de l'azote pour la production Agricole et Forestiere en Afrique. III eme conference de L'AABNF, 7–12 November 1988. ISRA Dakar, Senegal, pp 27–23
Rennie RJ, Dubetz S, Bole JB, Muendel HH (1982) Dinitrogen fixationt measured by15N isotope dilution in two Canadian soybean cultivars. Agron J 74:725–730
Vincent JM (1970) A manual for the practical study of root nodule bacteria. International Biological Programme Handbook no. 15, Blackwell, Oxford
Westermann DT, Kleinkopf GE, Porter LK, Loggett GE (1981) Nitrogen sources for bean seed production. Agron J 73:660–664
Zapata F, Danso SKA, Hardarson G, Fried M (1987a) Time course of nitrogen fixation in field-grown soybean using nitrogen-15 methodology. Agron J 79:172–176
Zapata F, Danso SKA, Hardarson G, Fried M (1987b) Time course of nitrogen fixation in field-grown faba bean using nitrogen-15 methodology. Agron J 79:505–509
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Kumaga, F., Danso, S.K.A. & Zapata, F. Time-course of nitrogen fixation in two bambara groundnut (Vigna subterranea L. Verdc.) cultivars. Biol Fert Soils 18, 231–236 (1994). https://doi.org/10.1007/BF00647672
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00647672