Summary
-
1.
The bilaterally paired ventral white cells (VWCs) of the buccal ganglion ofPleurobranchaea drive rhythmic motor output in the neural network controlling the movements of the buccal mass, which mediates both ingestion and egestion behaviors. The VWCs exert their action during minutes-long episodes of endogenously sustained depolarization and repetitive firing attended by pronounced action potential broadening (Gillette et al. 1980). In isolated preparations the ability to sustain such burst episodes is variable and somewhat labile (Gillette et al. 1980). We have assessed the ability of the intracellular messenger cyclic-3′,5′-adenosine monophosphate (cAMP), its non-hydrolyzable analogs and inhibitors of degradation to stimulate action potential broadening and bursting in the VWC.
-
2.
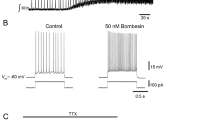
Intracellular injection of cAMP enhances progressive spike broadening (Figs. 1 and 2C) and stimulates spontaneous spiking activity and endogenous burst episodes in an apparent dose-dependent fashion (Fig. 2). Bath application of cAMP analogs or the phosphodiestrase inhibitor isobutylmethylxanthine also stimulates spike broadening and recurrent burst episodes in the intact neuron (Figs. 3 and 5) and induces the capacity to sustain prolonged bursts triggered by brief current stimulation in the isolated neuron cell body (Fig. 4).
-
3.
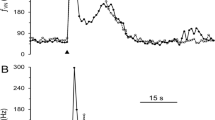
Work described in in the preceding paper (Gillette et al. 1982) suggested that regulation of (Ca++)i may affect progressive spike broadening in the VWC via a Ca++-activated K+ current (IK,Ca) and possibly via inward Ca++ current. The action of cAMP in enhancing spike broadening resembles that of agents and treatments that reduce (Ca++)i and/or suppress IK,Ca. Further, cAMP stimulation reverses a suppres sion of spike broadening caused by high Ca++ saline (Fig. 5) (which may increase [Ca++]i) and, conversely, that intracellular injection of a ‘high’ Ca++ buffer (Ca++-EGTA, 5 × 10−7 free Ca++) reverses the cAMP stimulation of spike broadening (Fig. 6).
-
4.
Stimulation with cAMP causes an abbreviation of the waveform and amplitude of the undershoots of single, unbroadened action potentials; this action resembles the effect of agents and treatments that reduce [Ca++]i and/or IK,Ca (Figs. 7, 8). This effect is reversed by intracellular injection of high Ca++ buffer (Fig. 9).
-
5.
These results suggest that one possible mechanism of action of cAMP is to reduce the ability or availability of intracellular free Ca++ to activate IK,Ca. A non-exclusive alternative is that cAMP enhances a Ca++ current. The evidence that both of these mechanisms could be regulated by an increase in the internal Ca++ buffering ability of the cell is discussed.
-
6.
The potential context of the VWC's behavioral function is one in which their command ability is induced by sensory or other input during arousal of some buccal behavior through neurotransmitter stimulation of endogenous cAMP.
Similar content being viewed by others
Abbreviations
- cAMP :
-
cyclic-3′,5′-adenosine monophosphate
- IBMX :
-
isobutylmethylxanthine
- CPT-cAMP :
-
8-p-chlorophenyl-thio-adenosine-3′,5′-cyclic monophosphoric acid
- VWC :
-
ventral white cell
References
Aldrich RW, Getting PA, Thompson SH (1979) Mechanism of frequency-dependent broadening of molluscan soma spikes. J Physiol (Lond) 291:531–544
Brunelli M, Castelucci V, Kandel ER (1976) Synaptic facilitation and behavioral sensitization inAplysia: possible role of serotonin and cyclic AMP. Science 194:1178–1180
Cedar H, Schwartz JH (1972) Cyclic adenosine monophosphate in the nervous system ofAplysia californica. II. Effect of serotonin and dopamine. J Gen Physiol 60:570–587
Eckert R, Tillotson D (1978) Potassium activation associated with intraneuronal free calcium. Science 200:437–439
Gillette R, Davis WJ (1977) The role of the metacerebral giant neuron in the feeding behavior ofPleurobranchaea. J Comp Physiol 116:129–159
Gillette R, Kovac MP, Davis WJ (1978) Command neurons receive synaptic feedback from the motor network they excite. Science 199:798–801
Gillette R, Gillette MU, Davis WJ (1980) Action potential broadening and endogenously sustained bursting are substrates of command ability in a feeding neuron ofPleurobranchaea. J Neurophysiol 43:669–685
Gillette R, Gillette MU, Davis WJ (1982) Substrates of command ability in a buccal neuron ofPleurobranchaea. I. Mechanisms of action potential broadening. J Comp Physiol 146:449–459
Gorman ALF, Thomas MV (1978) Changes in the intracellular concentration of free calcium ions in a pace-maker neurone, measured with the metallochromic indicator dye Arsenazo III. J Physiol (Lond) 275:357–376
Gorman ALF, Thomas MV (1980) Potassium conductance and internal calcium accumulation in a molluscan neurone. J Physiol (Lond) 308:287–313
Hidaka T, Osa T, Twarog BM (1967) The action of 5-HT onMytilus smooth muscle. J Physiol (Lond) 192:869–877
Junge D, Stevens CL (1973) Cyclic variation of potassium conductance in a burst-generating neurone inAplysia. J Physiol 235:155–181
Kaczmarek LK, Strumwasser F (1981) The expression of long lasting afterdischarge by isolatedAplysia bag cell neurons. J Neurosci 1:626–634
Klein M, Kandel ER (1978) Presynaptic modulation of voltagedependent Ca+2 current: Mechanism for behavioral sensitization inAplysia californica. Proc Natl Acad Sci USA 75:3512–3516
Kostyuk PG, Krishtal OA (1977) Effects of calcium and calciumchelating agents on the inward and outward current in the membrane of mollusc neurones. J Physiol (Lond) 270:569–580
Levitan I (1978) Adenylate cyclase in isolatedHelix andAplysia neuronal cell bodies: Stimulation by seroton and peptide-containing extract. Brain Res 154:404–408
Meyer RB Jr, Miller JP (1974) Analogs of cyclic AMP and cyclic GMP: General methods of synthesis and the relationship of structure to enzymic activity. Life Sci 14:1019–1040
Requena J, DiPolo R, Brinley FJ Jr, Mullins LJ (1977) The control of ionized calcium in squid axons. J Gen Physiol 70:329–353
Shimahara T, Tauc L (1977) Cyclic AMP induced by serotonin modulates the activity of an identified synapse inAplysia by facilitating the active permeability to calcium. Brain Res 127:168–172
Tillotson D (1979) Inactivation of Ca conductance dependent on entry of Ca ions in molluscan neurons. Proc Natl Acad Sci USA 76:1497–1500
Treistman SN, Levitan IB (1976) Alteration of electrical activity in molluscan neurones by cyclic nucleotides and peptide factors. Nature 261:62–64
Twarog BM (1966) Catch and the mechanism of action of 5-HT on molluscan muscle: A speculation. Life Sci 5:1201–1213
Twarog BM, Muneoka Y (1973) Calcium and the control of contraction and relaxation in a molluscan catch muscle. Cold Spring Harbor Symp Quant Biol 37:489–504
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Gillette, R., Gillette, M.U. & Davis, W.J. Substrates of command ability in a buccal neuron ofPleurobranchaea . J. Comp. Physiol. 146, 461–470 (1982). https://doi.org/10.1007/BF00609442
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00609442