Abstract
-
1.
Contraction of ipsilateral limb extensors during side-down roll tilt of the head, leading to selective stimulation of labyrinth receptors, is attributed to an increased discharge of excitatory vestibulospinal (VS) neurons (α-responses) and a decreased discharge of medullary inhibitory reticulospinal (RS) neurons (β-responses), both of which act on ipsilateral extensor motoneurons.
-
2.
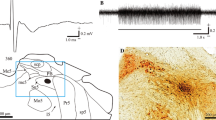
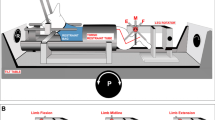
Experiments were performed in decerebrate cats, with the de-efferented gastrocnemius-soleus (GS) muscle fixed at a constant length, to find out whether Renshaw (R) cells linked with GS motoneurons responded to labyrinth stimulation elicited by head rotation, while the neck had been bilaterally deafferented. We hoped in this way to clarify the role and the mechanism by which these inhibitory interneurons act on limb extensor motoneurons during the vestibular reflexes.
-
3.
72.7% of the R-cells, disynaptically excited by group I volleys elicited by single shock stimulation of the GS nerve, weakly responded to head rotation at frequencies of 0.026–0.15 Hz and at a peak amplitude of 10°.
-
4.
For the frequency of head rotation of 0.026 Hz, ±10°C, most of the GS R-cells increased their firing rate during side-down head displacement (α-responses); some responses were related to head position, but others showed some phase lead or lag with respect to head position. The gain of the first harmonic of these unit responses was very low and corresponded on the average to 0.084±0.062, S.D. imp./s/deg, while the sensitivity corresponded to 2.14±2.35, S.D.%/deg (base frequency, 6.85±5.97, S.D.imp./s). These responses were attributed to the activity of VS neurons, the increased discharge of which during side-down head rotation exerts a weak excitatory influence on a limited number of GS motoneurons and, through their recurrent collaterals, on the related R-cells.
-
5.
The modulation of the firing rate of R-cells coupled with the GS motoneurons increased linearly by increasing the peak amplitude of displacement from 5° to 20° at the frequency of 0.026 Hz, so that the response gain remained almost unchanged.
-
6.
An increase in frequency of head rotation from 0.026 to 0.32 Hz at a fixed amplitude of 10°, thus changing the maximal angular acceleration from 0.26°/s2 to 41.7°/s2, reversed the response pattern of R-cells reported above. The resulting β-responses, which also showed some phase lead or lag with respect to head position, were attributed to vestibular activation of RS neurons. These neurons may directly excite the R-cells linked with the GS motoneurons, so that their reduced discharge during side-down head rotation would lead to a decrease in firing rate of the corresponding inhibitory interneurons. Interestingly, the response gain and sensitivity of these R-cells first decreased but then increased after reversal of the response pattern, by increasing frequency of head rotation. The critical frequency at which a cancellation of the response occurred before the response pattern was reversed, ranged between 0.051 and 0.15 Hz.
-
7.
In conclusion, R-cells linked with the GS motoneurons displayed an α-pattern of response during low frequency head rotation, suggesting that they were largely activated by the recurrent collaterals of the corresponding motoneurons driven by the VS volleys. The same R-cells, however, exhibited the opposite β-pattern of response at higher stimulus frequencies, thus behaving as if they were decoupled from their input motoneurons and controlled more directly by the medullary RS neurons. In these instances, activation of GS motoneurons during side-down head rotation could be attributed not only to an increased discharge of the excitatory VS neurons, but also to a reduced discharge of the R-cells driven by the medullary RS neurons. This system may thus contribute to the gain regulation of the EMG responses of hindlimb extensors to labyrinth stimulation.
Similar content being viewed by others
References
Anden N-E, Jukes MGM, Lundberg A, Byklický L (1966) The effect of DOPA on the spinal cord. 1. Influence on transmission from primary afferents. Acta Physiol Scand 67:373–386
Anderson JH, Blanks RHI, Precht W (1978) Response characteristics of semicircular canal and otolith systems in cat. I. Dynamic response of primary vestibular fibers. Exp Brain Res 32:491–507
Benecke R, Hellweg C, Meyer-Lohmann J (1974) Activity and excitability of Renshaw cells in non-decerebrate and decerebrate cats. Exp Brain Res 21:113–124
Benecke R, Meyer-Lohmann J, Guntau J (1976) Inverse changes in the excitability of Renshaw cells and α-motoneurones induced by interpositus stimulation. Pflügers Arch (Suppl) 365:40
Boyle R, Pompeiano O (1980) Reciprocal responses to sinusoidal tilt of neurons in Deiters' nucleus and their dynamic characteristics. Arch Ital Biol 118:1–32
Boyle R, Pompeiano O (1981) Convergence and interaction of neck and macular vestibular inputs on vestibulospinal neurons. J Neurophysiol 45:852–868
Boyle R, Pompeiano O (1984) Discharge activity of spindle afferents from the gastrocnemius-soleus muscle during head rotation in the decerebrate cat. Pflügers Arch 400:140–150
Cullheim S, Kellerth J-O (1978) A morphological study of the axons and recurrent axon collaterals of cat α-motoneurones supplying different functional types of muscle unit. J Physiol (Lond) 281:301–313
Denoth F, Magherini PC, Pompeiano O, Stanojević M (1979) Responses of Purkinje cells of the cerebellar vermis to neck and macular vestibular inputs. Pflügers Arch 381:87–98
Eccles JC, Fatt P, Koketsu K (1954) Cholinergic and inhibitory synapses in a pathway from motor axon collaterals to motoneurones. J Physiol (Lond) 126:524–562
Fernández C, Goldberg JM (1976) Physiology of peripheral neurons innervating otolith organs of the squirrel monkey. I. Response to static tilts and to long-duration centrifugal force. J Neurophysiol 39:970–984
Fernández C, Goldberg JM, Abend WK (1973) Response to static tilts of peripheral neurons innervating otolith organs of the squirrel monkey. J Neurophysiol 36:978–997
Goldberg JM, Fernández C (1975) Vestibular mechanisms. Ann Rev Physiol 37:129–162
Granit R, Haase J, Rutledge LT (1960) Recurrent inhibition in relation to frequency of firing and limitation of discharge rate of extensor motoneurones. J Physiol (Lond) 154:308–328
Haase J, van der Meulen JD (1961) Effects of supraspinal stimulation on Renshaw cells belonging to extensor motoneurones. J Neurophysiol 24:510–520
Haase J, Vogel B (1971) Die Erregung der Renshaw-Zellen durch reflektorische Entladungen der α-Motoneurone. Pflügers Arch 325:14–27
Haase J, Vogel B (1971) Direkte und indirekte Wirkungen supraspinaler Reizungen auf Renshaw-Zellen. Pflügers Arch 325:334–346
Henatsch H-D, Kaese H-J, Langrehr D, Meyer-Lohmann J (1961) Einflüsse des motorischen Cortex der Katze auf die Renshaw-Rückkopplungshemmung der Motoneurone. Pflügers Arch 274:51–52
Jankowska E, Lund S, Lundberg A, Pompeiano O (1968) Inhibitory effects evoked through ventral reticulospinal pathways. Arch Ital Biol 106:124–140
Koehler W, Windhorst U, Schmidt J, Meyer-Lohmann J, Henatsch H-D (1978) Diverging influences on Renshaw cell responses and monosynaptic reflexes from stimulation of capsula interna. Neuroscience Lett 8:35–39
Koizumi K, Ushiyama J, Brooks CMcC (1959) A study of reticular formation action on spinal interneurones and motoneurones. Jap J Physiol 9:282–303
Lindsay KW, Roberts TDM, Rosenberg JR (1976) Asymmetric tonic labyrinth reflexes and their interaction with neck reflexes in the decerebrate cat. J Physiol (Lond) 261:583–601
Llinás R, Terzuolo CA (1964) Mechanisms of supraspinal actions upon spinal cord activities. Reticular inhibitory mechanisms on alpha extensor motoneurons. J Neurophysiol 27:579–591
Loe PR, Tomko DL, Werner G (1973) The neural signal of angular head position in primary afferent vestibular nerve axons. J Physiol (Lond) 230:29–50
Lund S, Pompeinano O (1968) Monosynaptic excitation of alpha-motoneurons from supraspinal structures in the cat. Acta Physiol Scand 73:1–21
MacLean JB, Leffman H (1967) Supraspinal control of Renshaw cells. Exp Neurol 18:94–104
Magoun HW, Rhines R (1946) An inhibitory mechanism in the bulbar reticular formation. J Neurophysiol 9:165–171
Manzoni D, Pompeiano O, Srivastava UC, Stampacchia G (1983) Responses of forelimb extensors to sinusoidal stimulation of macular labyrinth and neck receptors. Arch Ital Biol 121:205–214
Manzoni D, Pompeiano O, Stampacchia G, Srivastava UC (1983) Responses of medullary reticulospinal neurons to sinusoidal stimulation of labyrinth receptors in decerebrate cat. J Neurophysiol 50:1059–1079
Peterson BP, Pitts NG, Fukushima K (1979) Reticulospinal connections with limb and axial motoneurons. Exp Brain Res 36:1–20
Pompeiano O (1975) Macular input to neurons of the spinoreticulocerebellar pathway. Brain Res 95:351–368
Pompeiano O (1975) Vestibulo-spinal relationships. In: Naunton RF (ed) The vestibular system. Academic Press, New York San Francisco London, pp 147–180
Pompciano O (1979) Neck and macular labyrinthine influences on the cervical spino-reticulocerebellar pathway. Progr Brain Res 50:501–514
Pompeiano O (1984) Recurrent inhibition. In: Davidoff RA (ed) Handbook of the spinal cord, vol 2–3. Marcell Dekker Inc, New York, pp 461–557
Pompeiano O, Wand P (1976) The relative sensitivity of Renshaw cells to static and dynamic changes in muscle length. Progr Brain Res 44:199–222
Pompeiano O, Wand P, Sontag K-H (1975) Response of Renshaw cells to sinusoidal stretch of hindlimb extensor muscles. Arch Ital Biol 113:205–237
Pompeiano O, Wand P, Sontag K-H (1975) The relative sensitivity of Renshaw cells to orthodromic group I a volleys caused by static stretch and vibration of extensor muscles. Arch Ital Biol 113:238–279
Pompeiano O, Wand P, Srivastava UC (1983) Labyrinthine influences on Renshaw cells coupled with gastrocnemius-soleus (GS) motoneurons. Proc XXIX Int Congr Physiol Sci, Sydney 15:85
Pompeiano O, Wand P, Srivastava UC (1984) Influence of Renshaw cells on the response gain of hindlimb extensor muscles to sinusoidal labryrinth stimulation. Pflügers Arch (in press)
Renshaw B (1946) Central effects of centripetal impulses in axons of spinal ventral roots. J Neurophysiol 9:191–204
Ross H-G, Cleveland S, Haase J (1972) Quantitative relation of Renshaw cell discharge to monosynaptic reflex height. Pflügers Arch 332:73–79
Ryall RW, Piercey MF, Polosa C, Goldfarb J (1972) Excitation of Renshaw cells in relation to orthodromic and antidromic excitation of motoneurons. J Neurophysiol 35:137–148
Schor RH, Miller AD (1981) Vestibular reflexes in neck and forelimb muscles evoked by roll tilt. J Neurophysiol 46:167–178
Schor RH, Miller AD (1982) Relationship of cat vestibular neurons to otolith-spinal reflexes. Exp Brain Res 47:137–144
Shimazu H, Precht W (1965) Tonic and kinetic responses of cat's vestibular neurons to horizontal angular acceleration. J Neuropysiol 28:991–1013
Vidal J, Jeannerod M, Lifschitz W, Levitan H, Rosenberg J, Segundo JP (1971) Static and dynamic properties of gravitysensitive receptors in the cat vestibular system. Kybernetik 9:205–215
Wand P, Pompeiano (1979) Contribution of different size motoneurons to Renshaw cell discharge during stretch and vibration reflexes. Progr Brain Res 50:45–60
Wand P, Pompeiano O, Fayein NA (1980) The relative sensitivity of different size motoneurons to group I a volleys caused by static stretch and vibration of extensor muscles. Arch Ital Biol 118:243–269
Windhorst U, Ptok M, Meyer-Lohmann J, Schmidt J (1978) Effects of conditioning stimulation of the contralateral n. ruber on antidromic Renshaw cell responses and monosynaptic reflexes. Pflügers Arch 373:R70
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Pompeiano, O., Wand, P. & Srivastava, U.C. Responses of Renshaw cells coupled with hindlimb extensor motoneurons to sinusoidal stimulation of labyrinth receptors in the decerebrate cat. Pflugers Arch. 403, 245–257 (1985). https://doi.org/10.1007/BF00583595
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00583595