Abstract
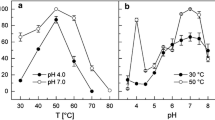
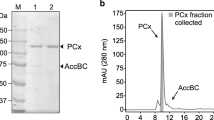
Phosphoenolpyruvate carboxylase (EC 4.1.1.31), which catalyzes the carboxylation of phosphoenolpyruvate to produce oxaloacetate was purified 465-fold from extracts of organotrophically grownThiobacillus novellus. Nondenaturing polyacrylamide gel electrophoresis (PAGE) of the purified enzyme revealed the presence of two bands after staining with Buffalo Black. Gels stained with Fast Violet B after incubation with PEP, HCO3 -, Mg2+ and acetyl CoA also showed two bands of activity with the faster moving the more active of the two. Sodium dodecylsulfate (SDS)-PAGE of the enzyme heated at 100°C for 5 min revealed the presence of three intensely stained bands of Mr 95 K, 51 K, and 28 K. However, electrophoresis of the enzyme heated for 2 min showed a single band of about 100 K, indicating that the preparation was likely homogeneous. The 51 K and 28 K subunits are thus products of the 95 K subunit. Gel filtration studies of the native enzyme yielded a Mr of 360 K. Therefore, the enzyme is a tetramer. The optimum pH in Tris buffer was 8.0, with Km for PEP 0.64 mM, HCO3 - 0.11 mM, and acetyl CoA a potent activator, 1.3 μM. A divalent cation best served by Mg2+ gave sigmoidal initial velocity plots. Hill plots of the data gave coefficients (nH) of 2.6. None of the metabolites tested, nucleotide triophosphates excepted, significantly affected enzyme activity. Binding studies with14C-labelled PEP revealed the binding of about 20 moles PEP per mole (360,000 g) of PEPC. Initial velocity studies suggest that the reaction is catalyzed by a random Bi Bi mechanism. Despite the lack of inhibition by certain metabolites, the enzyme's function is probably anaplerotic.
Similar content being viewed by others
References
Andreo CS, Gonzales DH & Iglesias AA (1987) Higher plant phosphoenolpyruvate carboxylase: structure and regulation. FEBS Lett. 213: 1–8
Andrews P (1970) Estimation of molecular size and molecular weights of biological compounds by gel filtration.In: Glick D (Ed) Methods of Biochemical Analysis, Vol 18 (pp 1–153). Interscience Publishers, Toronto
Atkinson DE (1968) The energy charge of the adenylate pool as a regulatory parameter. Interaction with feedback modifiers. Biochemistry, 7: 4030–4034
Bailey JL (1967) Techniques in Protein Chemistry (2nd ed.) Elsevier Publishing Co., New York, 345 pp
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72: 248–254
Canovas JL & Kornberg HL (1969) Phosphoenolpyruvate carboxylase fromEscherichia coli. In: Lowenstein JM (Ed) Methods in Enzymology, Vol 13 (pp 288–292). Academic Press, New York
Charles AM & Willer DW (1984) Pyruvate carboxylase fromThiobacillus novellus: properties and possible function. Can. J. Microbiol. 30: 532–539
Cleland WW (1963) The kinetics of enzyme catalyzed reactions with two or more substrates or products I. Nomenclature and rate equations. Biochem Biophys Acta 67: 104–137
Coombs J, Maw SL & Baldry CW (1974) Metabolic regulation in C4 photosynthesis: PEP carboxylase and energy charge. Planta 117: 279–292
Cornish-Bowden A (1979) Fundamentals of Enzyme Kinetics (pp 25–28). Butterworth and Co. Ltd., Sydney
Gekko K & Timasheff SN (1981) Mechanism of protein stabilization by glycerol: preferential hydration in glycerol water mixtures. Biochemistry 20: 4667–4676
Hay RW (1976) Metal ion-catalyzed decarboxylations of biological interest. In: Siegel H (Ed) Metal Ions in Biological Systems, Vol 5 (pp 127–172). M. Dekker, New York
Kornberg HL (1966) Anaplerotic sequences and their role in metabolism. In: Campbell PN & Greville GD (Eds)_ Essays in Biochemistry, Vol 2 (pp 1–31). Academic Press, New York
Laemmli UK (1970) Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 227: 680–685
Maeba P & Sanwal BD (1969) Phosphoenolpyruvate carboxylase ofSalmonella. Some chemical and allosteric properties. J. Biol. Chem. 244: 2549–2557
McCarthy JT & Charles AM (1974) CO2 fixation by the facultative autotrophThiobacillus novellus during autotrophy heterotrophy interconversions. Can J Microbiol 20: 1577–1584
Miziorko HM, Nowak T & Mildvan AS (1974) Spinach leaf phosphoenolpyruvate carboxylase: purification, properties and kinetic studies. Arch Biochem Biophys 163: 378–389
Mori M & Shiio I (1985) Purification and some properties of phosphoenolpyruvate carboxylase fromBrevibacterium flavum and its aspartate-overproducing mutant. J Biochem 97: 1119–1128
Morikawa M, Izui K, Taguchi M & Katsuki H (1980) Regulation ofEscherichia coli phosphoenolpyruvate carboxylase by multiple effectorsin vivo. J Biochem 87: 441–449.
Mukerji SK (1977) Corn leaf phosphoenolpyruvate carboxylases. The effect of divalent cations on activity. Arch Biochem Biophys 182: 352–359
Nguyen TT, Ngam-ek A, Jenkins J & Grover S (1988) Metal ion interactions with phosphoenolpyruvate carboxylase fromCrassula argentea andZea mays. Plant Physiol 86: 104–107
Nielsen BL & Brown LR (1984) The basis for colored silver-protein complex formation in stained polyacrylamide gels. Anal Biochem 141: 311–315
Nimmo GA, McNaughton GAL, Fewson CA, Wilkins MB & Nimmo HG (1987) Changes in the kinetic properties and phosphorylation state of phosphoenolpyruvate carboxylase inZea mays leaves in response to light and dark. FEBS Lett 213: 18–22
O'Leary M (1982) Phosphoenolpyruvate carboxylase: an enzymologist's view. Annu Rev Plant Physiol 33: 297–315
Owttrim GW & Colman B (1986) Purification and characterization of phosphoenolpyruvate carboxylase from a cyanobacterium. J. Bacteriol 168: 207–212
Paulus HC (1969) A rapid and sensitive method for determining the binding of radioactive ligands to proteins. Anal Biochem 32: 91–100
Sato F, Koizumi N & Yamada Y (1988) Purification and characterization of phosphoenolpyruvate carboxylase of photomixotrophically cultured green tobacco cells. Plant Cell Physiol 29: 329–337
Schwitzguebel J-P & Ettlinger L (1979) Phosphoenolpyruvate carboxylase fromAcetobacter aceti. Arch Microbiol 122: 109–115
Scrutton MC, Olmstead MR & Utter MF (1969) Pyruvate carboxylase from chicken liver. In: Lowenstein JM (Ed) Methods in Enzymology, Vol 13 (pp 235–250). Academic Press, New York
Scrutton MC & Fung CH (1972) Pyruvate carboxylase from chicken: effects of sulfate and other anions on catalytic activity and structural parameters. Arch Biochem Biophys 150: 636–647
Scrutton MC & Fatebene F (1975) An assay system for localization of pyruvate and phosphoenolpyruvate carboxylase activity on polyacrylamide gels and its application to detection of these enzymes in tissue and cell extracts. Anal Biochem 69: 247–260
Scrutton MC (1978) Fine control of the conversion of pyruvate (phosphoenolpyruvate) to oxaloacetate in various species. FEBS Lett 89: 1–9
Selinioti E, Nikolopoulos D & Manetas Y (1987) Organic cosolutes as stabilizers of phosphoenolpyruvate carboxylase in storage: an interpretation of their action. Aust J Plant Physiol 14: 203–210
Singal HR & Singh R (1986) Purification and properties of phosphoenolpyruvate carboxylase from immature pods of chickpea (Cicer arietinum L). Plant Physiol 80: 369–373
Stamatakis K, Gavalas NA & Manetas Y (1988) Organic cosolutes increase the catalytic efficiency of phosphoenolpyruvate carboxylase fromCynodon dactylon (L) pers., apparently through self-association of the enzymic protein. Aust J Plant Physiol 15: 621–631
Suzuki I & Werkman CH (1958) Chemoautotrophic carbon dioxide fixation by extracts ofThiobacillus thiooxidans. I. Formation of oxalacetic acid. Arch Biochem Biophys 76: 103–111
Utter MF & Kolenbrander HM (1972) Formation of oxaloacetate by CO2 fixation on phosphoenolpyruvate. In: Boyer PD (Ed) The Enzymes, Vol 6 (pp 117–136). Academic Press, New York
Wedding RH & Black MK (1988) Role of magnesium in the binding of substrate and effectors to phosphoenolpyruvate carboxylase from a CAM plant. Plant Physiol 87: 443–446
Willeford KO, Wu M-X, Meyer CR & Wedding RT (1990) The role of oligomerization in regulation of maize phosphoenolpyruvate carboxylase activity. Biochem Biophys Res Comm 168: 778–785
Yoshinaga T, Izui K & Katsuki H (1970) Purification and molecular properties of allosteric phosphoenolpyruvate carboxylase fromEscherichia coli. J Biochem 68: 747–750
Author information
Authors and Affiliations
Additional information
Supported by an operating grant from NSERC to AMC.
Rights and permissions
About this article
Cite this article
Charles, A.M., Sykora, Y. Purification and characterization of the phosphoenolpyruvate carboxylase from the facultative chemolithotrophThiobacillus novellus (ATCC 8093). Antonie van Leeuwenhoek 62, 155–165 (1992). https://doi.org/10.1007/BF00582575
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00582575