Abstract
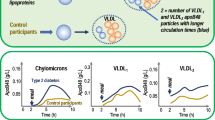
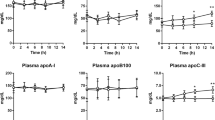
We have previously demonstrated alterations in apolipoprotein B-48 metabolism in the post-prandial state in patients with non-insulin-dependent diabetes mellitus. This study investigates the relationship between hypertriglyceridaemia and post-prandial lipoprotein metabolism. Four groups of patients were examined: non-insulin-dependent diabetic patients, with normal serum triglyceride levels (serum triglyceride <2.1 mmol l−1; haemoglobin HbA1c 5.5%±0.4%); poorly controlled, non-insulin-dependent diabetic patients with hypertriglyceridaemia (serum triglyceride >2.1 mmol 1−1; HbA1c 8.8%±0.9%); nondiabetic subjects with serum triglycerides <2.1 mmoll−1; and non-diabetic subjects with hypertriglyceridaemia (serum triglyceride>2.1 mmol l−1). Subjects were studied fasting and following a high-fat meal (1300 kcal). The triglyceride-rich lipoprotein fraction was isolated by ultracentrifugation (d<1.006 g ml−1). Apoprotein B-48, apoprotein B-100 and apoprotein E were separated on 4%–15% gradient gels and quantified as a percentage of the fasting concentration by densitometric scanning. Triglyceride-rich lipoprotein apolipoprotein B-48 and apolipoprotein B-100 post-prandial profiles demonstrated a maximum increase either at 2 h or rising still further to a peak at 6 h before falling in the diabetic groups and hypertriglyceridaemic non-diabetic subjects when compared with the normotriglyceridaemic control subjects whose levels decreased after 2 h (P<0.05). A significantly different triglyceride-rich lipoprotein apolipoprotein E profile was also exhibited by the diabetic patients (P<0.05). Levels of triglyceride-rich lipoprotein, cholesterol, triglyceride, total protein and apoprotein B were elevated in the hypertriglyceridaemic subjects, both diabetic and non-diabetic. These results indicate that hypertriglyceridaemia is associated with altered metabolism and composition of post-prandial triglyceride-rich lipoprotein particles in both poorly controlled diabetic and non-diabetic subjects.
Similar content being viewed by others
References
Dunn FL, Hyperlipidemia in diabetes mellitus. Diabetes Metab Rev 6:47–61, 1990
Howard BV, Reitman JS, Vasquez B, Zech L, Very-low-density lipoprotein triglyceride metabolism in non-insulin-dependent diabetes mellitus. Relationship to plasma insulin and free fatty acids. Diabetes 32:271–276, 1983
Taskinen M-R, Beltz WF, Harper I, Effects of NIDDM on verylow density lupoprotein triglyceride and apolipoprotein B metabolism: studies before and after sulphonylurea therapy. Diabetes 35:1268–1277, 1986
Brunzell JD, Porte Jr D, Bierman EL, Abnormal lipoprotein lipase-mediated plasma triglyceride removal in untreated diabetes mellitus associated with hypertriglyceridemia. Metabolism 28:901–907, 1979
Simsolo RB, Ong JM, Saffari B, Kern PA, Effect of improved diabetes control on the effect of lipoprotein lipase in human adipose tissue. J Lipid Res 33:89–95, 1992
Zilversmit DB, Atherogenesis: a postprandial phenomenon. Circulation 3:473–485, 1979
Wilson DE, Chan I-F, Buchi KN, Horton SC, Postchallenge plasma lipoprotein retinoids: chylomicrons remnants in endogenous hypertriglyceridemia. Metabolism 34:551–558, 1985
Curtin A, Deegan P, Owens D, Collins P, Johnson A, Tomkin GH, Alterations in apolipoprotein B48 in the post-prandial state in NIDDM. Diabetologia 37:1259–1264, 1994
Markwell MAK, Haas SM, Bieber LL, Tolbert NE, A modification of the Lowry procedure to simplify protein determination in membrane and lipoprotein samples. Anal Biochem 87:206–210, 1978
Adell K, Ogbonna G, Rapid purification of DNA from whole blood for potential application in clinical chemistry laboratories. Clin Chem 36:261–264, 1990
Hixson JE, Vernier DT, Restriction isotyping of human apolipoprotein E by gene amplification and cleavage withHha 1. J Lipid Res 31:545–548, 1990
Cohn JS, McNamara JR, Cohn SD, Ordovas JM, Schaefer EJ, Plasma apolipoprotein changes in the triglyceride-rich lipoprotein fraction of human subjects fed a fat-rich meal. J Lipid Res 29:925–936, 1988
Patsch JR, Prasad S, Gotto Jr AM, Patsch W, High density lipoprotein 2: relationship of the plasma levels of this lipoprotein species to its composition, to the magnitude of postprandial lipaemia and to the activities of lipoprotein lipase and hepatic lipase. J Clin Invest 80:341–347, 1987
Karpe F, Bard J-M, Steiner G, Carlson LA, Fruchart J-C, Hamsten A, HDLs and alimentary lipaemia: studies in men with previous myocardial infarction at a young age. Arteriosclerosis Thromb 13:11–22, 1993
Shue WHH, Shieh SM, Fue MMT, Shen DDC, Jeng CY, Chen YDI, Reaven GM, Insulin resistance, glucose intolerance, and hyperinsulinemia: hypertriglyceridemia versus hypercholesterolemia. Arteriosclerosis Thromb 13:367–370, 1993
Zimmet PZ, Hyperinsulinemia — how innocent a bystander? Diabetes Care 16:56–70, 1993
Taskinen M-R, Hyperlipidaemia in diabetes. Baillieres Clin Endocrinol Metabol 4: 743–775, 1990
Tan KCB, Cooper MB, Ling KLE, Postprandial lipoprotein metabolism in type 2 diabetes mellitus. Diabetologia 35:168, 1992
Georgopoulos A, Margolis S, Bachorik P, Kwiterovich PO, Effect of improved glycaemic control on the response of plasma triglycerides to ingestion of a saturated fat load in normotriglyceridaemic and hypertriglyceridaemic diabetic subjects. Metabolism 37:866–872, 1988
Cohn JS, Johnson EJ, Millar JS, Cohn SD, Milne RW, Marcel YL et al, Contribution of apo B-48 and apo B-100 triglyceride-rich lipoproteins (TRL) to postprandial increases in the plasma concentration of TRL triglycerides and retinyl esters. J Lipid Res 34:2033–2040, 1993
Nestel PJ, Billington T, Fidge NH, Slower removal of intestinal apolipoprotein B-48 than of apolipoprotein B-100 in severely hypertriglyceridemic subjects. Biochem Biophys Acta 751:422–427, 1983
Karpe F, Steiner G, Uffelman K, Olivecrona T, Hamsten A, Postprandial lipoproteins and progression of coronary atherosclerosis. Atherosclerosis 106:83–97, 1994
Martins IJ, Sainsbury AJ, Mamo JCL, Redgrave TG, Lipid and apolipoprotein B48 transport in mesenteric lymph and the effect of hyperphagia on the clearance of chylomicron-like emulsions in insulin-deficient rats. Diabetologia 37:238–247, 1994
Young NL, Lopez DR, McNamara DJ, Contributions of absorbed dietary cholesterol and cholesterol synthesized in small intestine to hypercholesterolemia in diabetic rats. Diabetes 37; 1151–1156, 1988
O'Meara N, Devery R, Owens D, Collins P, Johnson A, Tomkin GH. Cholesterol metabolism in the alloxan-induced diabetic rabbit. Diabetes 39:626–633, 1990
Weintraub MS, Eisenberg S, Breslow JL, Dietary fat clearance in normal subjects is regulated by genetic variation in apolipoprotein E. J Clin Invest 80:1571–1577, 1987
Miettinen TA, Impact of apo E phenotype on the regulation of cholesterol metabolism. Ann Med 23:181–186, 1991
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Curtin, A., Deegan, P., Owens, D. et al. Intestinally derived lipoprotein particles in non-insulin-dependent diabetic patients with and without hypertriglyceridaemia. Acta Diabetol 32, 244–250 (1995). https://doi.org/10.1007/BF00576257
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00576257