Summary
The safety and tolerance of single oral doses of a new angiotensin converting enzyme (ACE) inhibitor, trandolapril have been examined in 90 healthy male volunteers, in a randomised, double blind, placebo-controlled study. The subjects were divided into 10 groups, each of 9 subjects and treatments (6 subjects on trandolapril and 3 on placebo per group) were allocated by unbalanced randomisation. Ten single, increasing oral doses were tested: 0.125, 0.25, 0.5, 1, 2, 4, 8, 16, 24 and 32 mg. The assessment criteria were clinical (monitoring of blood pressure, heart and respiratory rate, electrocardiogram, temperature and evaluation of behaviour and side effects) and routine laboratory tests.
Blood pressure did not fall except for a slight drop in diastolic pressure during the first 4 h following the 32-mg dose. However, although an effect of the compound cannot be excluded, the reduction in blood pressure may have reflected intersubject variability. No orthostatic hypotension was observed. There was no change in the other vital signs, and in particular no increase in heart rate was observed. No serious adverse effect was encountered.
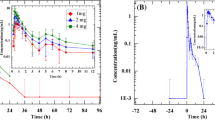
The pharmacological activity of the compound was studied by assaying plasma ACE activity. Inhibition of ACE was linearly dose-dependant from 0 (placebo) to 2 mg, and above that dose, the inhibition was nearly total. ACE activity was markedly reduced within 30 min after administration of trandolapril, and maximal inhibition was observed from 2–4 h onwards, lasting for up to 24 h after dosing. For doses above 2 mg, inhibition was still 40% of the basal activity on Day 8 after dosing.
Similar content being viewed by others
References
Ajayi AA, Lees KR, Reid JC (1986) Effects of angiotensin converting enzyme inhibitor, perindopril, on autonomic reflexes. Eur J Clin Pharmacol 30: 177–182
Biollaz J, Burnier M, Turini GA, Brunner DB, Porchet M, Gomez HJ, Jones KH, Ferber F, Abrams WB, Gavras H, Brunner HR (1981) Three long-acting-converting enzyme inhibitors: Relationship between plasma converting-enzyme activity and response to angiotensin I. Clin Pharmacol Ther 29: 665–670
Brunner DB, Desponds G, Biollaz J, Keller I, Ferber F, Gavras H, Brunner HR, Schelling JL (1981) Effect of a new angiotensin converting enzyme inhibitor MK 421 and its lysine analogue on the components of the renin system in healthy subjects. Br J Clin Pharmacol 11: 461–467
Brunner HR, Nussberger J, Waeber B (1985) The present molecules of converting enzyme inhibitors. J Cardiovasc Pharmacol 7: S2-S11
Burnier M, Turini GA, Brunner HR, Porchet M, Kruithof D, Vukovich RA, Gavras H (1981) RHC 3659: a new orally active angiotensin converting enzyme inhibitor in normal volunteers. Br J Clin Pharmacol 11: 461–467
Fasanella d'Amore T, Bussien JP, Nussberger J, Waeber B, Turini GA, Brunner HR, Kler L, Francis RJ (1987) Effects of single dose of the converting enzyme inhibitor cilazapril in normal volunteers. J Cardiovasc Pharmacol 9: 26–31
Harjanne A (1984) Automated kinetic determination of angiotensin-converting enzyme in serum. Clin Chem 30: 901–902
Imai Y, Abe K, Seino M, Haruyama T, Tajima J, Sato M, Goto T, Hiwatari M, Kasai Y, Yoshimaga K, Sekino H (1982) Attenuation of pressor responses to norepinephrine and pitressin and potentiation of pressor response to angiontensin II by captopril in human subjects. Hypertension 4: 444–451
Jacob des Combes B, Turini GA, Brunner HR, Porchet M (1983) CGS 13945: A new orally active angiotensin converting enzyme inhibitor in normal volunteers. J Cardiovasc Pharmacol 5: 511–516
Manhem PJO, Ball SG, Morton JJ, Murray GD, Leckie BJ, Fraser R, Robertson JIS (1985) A dose response study of HOE 498, a new non sulphydryl converting enzyme inhibitor, on blood pressure, pulse rate and the renin-angiotensin-aldosterene system in normal man. Br J Clin Pharmacol 20: 27–35
Mc Gregor GA, Markandu ND, Roulston JE, Jones JC (1979) Essentiel hypertension: Effect of an oral inhibitor of converting enzyme. Br Med J 2: 1106–1109
Mc Gregor GA, Markandu ND, Bayliss J, Roulston JE, Squires M, Morton I (1981) Non-sulfhydril containing angiotensin converting enzyme inhibitor (MK 421): Evidence for role and renin system in normotensine subjects. Br Med J 283: 401–403
Ogihara T, Nakamaru M, Higaki J, Kumahara Y (1987a) Pharmacodynamics and pharmacokinetics of a new orally active angiotensin I converting enzyme inhibitor, alacepril (DU 1219) in normal subjects and hypertensive patients. Curr Ther Res 41: 492–508
Ogihara T, Nakamaru M, Higaki J, Kumahara Y, Hamano Y, Minamino T, Nakamura N (1987b) Phase I study of a new orally active angiotensin — I — converting enzyme inhibitor, delapril. Curr Ther Res 41: 809–822
Rakhit A, Hurley ME, Tipnis V, Coleman J, Rommel A, Brunner HR (1986) Pharmacokinetics and pharmacodynamics of pentopril: a new angiotensin converting enzyme inhibitor in humans. J Clin Pharmacol 26: 156–164
Richer C, Thuillez C, Desmichels D, Giudicelli JF (1986) Effects hémodynamiques périphériques et biologiques du perindopril chez le volontaire sain. Relation dose-effet. Arch Mal Coeur 79: 913–918
Vierhapper H, Witte P, Waldhausl W (1982) Unchanged pressor effect of norepinephrine in normal man following the oral administration of two angiotensin converting enzyme inhibitors, captopril and HOE 498. J Hypertens 4: 9–11
Wellstein A, Essig J, Belz GG (1987) Inhibition of angiotensin I response by cilazapril and its time course in normal volunteers. Clin Pharmacol Ther 41: 639–644
Witte PU, Irmisch R, Hajdu P, Metzger H (1984b) Pharmacokinetics and pharmacodynamics of a novel orally active angiotenin-converting enzyme inhibitor (HOE 498) in healthy volunteers. Eur J Clin Pharmacol 27: 577–581
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Patat, A., Surjus, A., Le Go, A. et al. Safety and tolerance of single oral doses of trandolapril (RU 44.570), a new angiotensin converting enzyme inhibitor. Eur J Clin Pharmacol 36, 17–23 (1989). https://doi.org/10.1007/BF00561017
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00561017