Summary
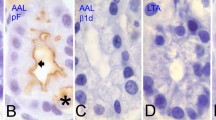
The localization of complex carbohydrates in the Golgi apparatus, secretory granules and plasmalemma of mouse parotid acinar cells was studied using the fracture-labelling method. The hexose residues of glycoconjugates were identified using ferritin conjugated with Wheat Germ Agglutinin (WGA-), Ricinnus Communis Agglutinin II (RCA-II-), Phaseolus Vulgaris Agglutinin (PHA-) and Limulus Polyphemus Agglutinin (LPA-). We found that the tracture-labelling method allows not only the labelling of membrane faces but also analysis of the compartment's content that is exposed during the fracturing of the tissue. Our results revealed differences in the hexose residues located in the Golgi apparatus, secretory granules and the apical and lateral plasmalemma. Numerous binding sites for WGA-, PHA-and RCA-II-ferritin were demonstrable in the Golgi apparatus. In secretory granules, the WGA-and RCA-II-ferritin binding sites were most numerous, while LPA-ferritin binding sites were very rate. The density of the binding sites for PHA-ferritin showed considerable variation in secretory granules. The apical plasmalemma exhibited a high density of binding sites for all of the lectins used. In the lateral plasmalemma, LPA-ferritin was not bound, and there were fewer binding sites for WGA-, RCA-H-and PHA-ferritin.
Similar content being viewed by others
References
Barbosa MLF, Pinto da Silva P (1983) Restriction of glycolipids to the outer half of a plasma membrane: Concavalin — A labelling of membrane halves in Acanthamoeba castellanii. Cell 33:959–966
Bennet G, Leblond CP (1977) Biosynthesis of the glycoproteins present in plasma membrane, lysosomes and secretory materials, as visualized by radioautography. Histochem J 9:393–417
Bennett B, Kan FWK, O'Shaughnessy D (1981) The site of incorporation of sialic acid residues into glycoproteins and the subsequent fates of these molecules into various rat and mouse cell types as shown by autoradiography after injection of (H3) N-acetyl mannosamine: observations in tissues other than liver. J Cell Biol 88:16–28
Berger EG (1985) Mini review: How Golgi-associated glycosylation works. Cell Biol Int Rep 9/5:407–417
Bergman JE, Tokuyasu KT, Singer SJ (1981) Passage of an integral membrane protein, the vesicular stomatitis virus glycoprotein, though the Golgi apparaturs en route to the plasma membrane. Proc Natl Acad Sci USA 78:1746–1750
Branton D (1971) Freeze-etching studies of membrane structure. Philos Trans R Soc Lond B 261L133–138
Branton D, Bullivant S, Gilula NB, Karnovsky MJ, Moor H, Mühlethaler K, Nortcote DH, Packer L, Satir B, Satir P, Speth V, Stachlin LA, Steere RL, Weinstein RS (1975) Freeze-etching nomenclature. Science 190:54–56
Camilli P, Peluchetti LD, Meldolesi J (1976) Dynamic changes of the luminal plasmalemma in stimulated parotid acinar cells. J Cell Biol 70:59–74
Casceiri AM, Somberg EW (1983) Two-dimensional electrophoretic analysis of secretory-granule, granule-membrane and plasmamembrane proteins of rat parotid cells. Cell Tissue Res 234:93–108
Castle JD, Jamieson JD, Palade GE (1972) Radioautographic analysis of the secretory process in the parotid acinar cell of the rabbit. J Cell Biol 53:290–311
Haddad A, Brasileiro ILG (1983) Synthesis of membrane and secretory glycoproteins by the acinar pancreatic cells as visualized by radioautography. Cell Tissue Res 233:197–207
Hynes RO (1979) Proteins and glycoproteins. In: Surfaces of nomal and malignant cells. John Wiley and Sons, Chichester, New York, pp 103–148
Iversen JM, Kauffman DL, Keller PJ, Robinovitch M (1985) Isolation and partial characterization of two populations of secretoty granules from rat parotid glands. Cell Tissue Res 240:441–447
Kauffman DL, Keller PJ (1979) The basic proline-rich proteins in human parotid saliva from a single subject. Arch Oral Biol 24:249–256
Keller PJ, Robinovitch M, Iversen J, Kauffman DL (1975) The protin composition of rat parotid saliva and secretory granules. Biochim Biophys Acta 379:562–570
Mendel ID, Thompson JRH, Allison SA (1965) Studies on the mucoproteins of human parotid saliva. Arch Oral Biol 10:499–507
Meldolesi J, Ceccarelli B (1981) Exocytosis and membrane recycling. Philos Trans R Soc Lond B 296:55–65
Pierce M, Turley EA, Roth S (1980) Cell surface glycosyltransferase activities. Int Rev Cytol 65:1–47
Pinkstaff CA (1980) The cytology of salivary glands. Int Rev Cytol 63:141–261
Pinto da Silva P, Parkinson C, Dwyer N (1981a) Fracturelabel: Cytochemistry of freeze-fracture faces in the erythrocyte membrane. Proc Natl Acad Sci USA 78/1:343–347
Pinto da Silva P, Parkinson C, Dwyer N (1981b) Freeze-Fracture cytochemistry: Thin sections of cell and tissues after labelling of fracture faces. J Histochem Cytochem 29:917–928
Pollac L, Atkinson PH (1983) Correlation of glycosilation forms with position in amino acid sequence. J Cell Biol 97:293–300
Robbins PW, Hubbard SC, Turco SJ, Wirth DF (1977) Proposal for a common oligosaccharide intermediate in the synthesis of membrane glycoproteins. Cell 12:893–900
Ronzio RA, Kronquist KE, Lewis DS, Macdonald RJ, Mohrlok SH, O'donnell JJ (1978) Glycoprotein synthesis in the adult rat pancreas. IV. Subcellular distribution of membrane glycoproteins. Biochem Biophys Acta 508:65–84
Roth J, Binder M (1978) Coloidal gold, ferritin and peroxidase as markers for electron microscopic double labelling lectin techniques. J Histochem Cytochem 26:163–169
Roth J, Lucocq JM, Berger EG, Paulson JC, Watkins WM (1984) Terminal glycosilation is compartmentalized in the Golgi apparatus. J Cell Biol 99:229a
Schulte BA, Spicer SS (1983) Light microscopic detection of sugar residues in glycoconjugates of salivary glands and the pancreas with lectin-horseredish peroxidase conjugates. Histochem J 15:1217–1238
Schulte BA, Spicer SS, Miller RL (1985) Lectin histochemistry of secretory and cell-surface glycoconjugates in the ovine submandibular gland. Cell Tissue Res 240:57–66
Sturgess J, Moscarello M, Schachter H (1978) The structure and biosynthesis of membrane glycoproteins. Curr Top Membr Transp 11:16–63
Suzuki S, Tsuyama S, Murata F (1982) Post-Embedding staining of rat gastric mucous cells with lectins. Histochemistry 73:563–575
Torrisi MF, Pinto da Silva P (1984) Compartmentalization of intracellular membrane glycocomponents is revealed by fracturelabel. J Cell Biol 98:29–34
Wallach D (1982) The secretory granule of the parotid gland. Poisner and Trifaro (eds) The secretory granule. Elsevier Biomedical Press, Amsterdam, pp 247–276
Wallach D, Kirshner N, Schramm M (1975a) Non-parallel Transport of membrane proteins and content proteins during assembly of the secretory granula in rat parotid gland. Biochim Biophys Acta 357:87–105
Wallach D, Tessler R, Schramm M (1975b) The proteins of the content of the secretory granules of the rat parotid gland. Biochim Biophys Acta 382:552–564
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Jezernik, K., Pipan, N. Lectin-binding pattern in parotid acinar cells. Histochemistry 85, 515–521 (1986). https://doi.org/10.1007/BF00508434
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00508434