Summary
-
1.
Serotonin (5-HT) induces inositol phosphate production and the efflux of 45Ca2+ in a smooth muscle cell line (A7r5) derived from rat aorta. These effects were pharmacologically characterised and compared to data obtained in radioligand binding studies performed with the 5-HT2 ligand [3H]ketanserin in rat brain cortex membranes.
-
2.
5-HT causes in increase in the levels of inositol trisphosphate (InsP3), inositol bisphosphate (InsP2) and inositol phosphate (InsP1). InsP3 production was rapid and transient whereas InsP1 accumulated in a time and concentration dependent manner. The 5-HT stimulated InsP1 accumulation (pEC50=6.48) was potently and competitively inhibited by the 5-HT2 specific antagonists, pirenperone and ketanserin, whereas antagonists of other 5-HT receptors were active only at high concentrations. There was a significant correlation between inhibition of 5-HT stimulated InsP1 accumulation and 5-HT2 binding (r=0.98, P=0.0035).
-
3.
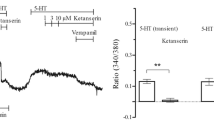
5-HT stimulated the efflux of 45Ca2+ from preloaded cells with a pEC50 of 7.59. The rank order of potency for agonist induced Ca2+ efflux, 5-HT > α-methyl-5-HT > 1-methyl-5-HT > RU 24969 (5-methoxy-3[1,2,3,6-tetrahy-dropyridin-4-yl]-1-H indole) > 8-OH-DPAT (8-hydroxy-2-(di-n-propylamino)tetralin) > 5-CT (5-carboxamidotryptamine) is typical for a 5-HT2 receptor mediated event. The effect of 5-HT was competitively blocked by ketanserin (pA2=8.22). There was a very good correlation (r=0.94; P=0.0001) between antagonism of Ca2+ release and 5-HT2 binding as measured using a series of 13 antagonists.
-
4.
The values obtained with selective agonists and antagonists exclude that the 5-HT mediated effects described here involve 5-HT1A, 1B, 1C, 5-HTM receptors, α-or β-adrenoceptors. The data suggests that 5-HT mobilises intracellular Ca2+ via the production of InsP3 in this smooth muscle cell line, an effect mediated by a 5-HT2 receptor.
Similar content being viewed by others
References
Affolter H, Erne P, Bürgisser E, Pletscher A (1984) Ca2+ as messenger of 5-HT2 receptor stimulation in human blood platelets. Naunyn-Schmiedeberg's Arch Pharamcol 325:337–342
Aghajanian GK (1981) The modulatory role of serotonin at multiple receptors in brain. In: Jacobs BL Gelperin A (eds) Serotonin neurotransmission and behaviour. MIT Press, Cambridge MA, pp 156–185
Alexander RW, Brock TA, Gimbrone MA, Ritenhouse SE (1985) Angiotensin increases inositol trisphosphate and calcium in vascular smooth muscle. Hypertension 7:447–451
Arunlakshana O, Schild HO (1959) Some quantitative uses of drug antagonists. Br J Pharmacol Chemother 14:48–58
Aub DL, Putney JW (1984) Metabolism of inositol phosphates in parotid cells: Implication for the pathway of the phosphoinositide effect and for the possible messenger role of inositol trisphosphate. Life Sci 34:1347–1355
Berridge MJ (1978) Phosphatidylinositol metabolism and calcium gating in a 5-HT receptor system. In: Birdsall NJM (ed) Drug receptors and their effectors. MacMillan Press, New York, pp 75–85
Berridge MJ (1983) Rapid accumulation of inositol trisphosphate reveals that agonists hydrolyse polyphosphoinositides instead of phosphatidyl inositol. Biochem J 212:849–858
Berridge MJ (1984) Inositol trisphosphate and diacylglycerol as second messengers. Biochem J 220:345–360
Berridge MJ, Dawson MRC, Downes CP, Heslop JP and Irvine RF (1983) Changes in the levels of inositol phosphates after agonistdependent hydrolysis of membrane phosphoinositides. Biochem J 212:473–482
Berridge MJ, Downes CP, Hanley MR (1982) Lithium amplifies agonist dependent phosphatidylinositol responses in brain and salivary glands. Biochem J 206:587–595
Brown E, Kendall DA, Nahorski SR (1984) Inositol phospholipid hydrolysis in rat cerebral cortex slices. I. Receptor characterisation. J Neurochem 42:1379–1387
Burgess GM, Godfrey PP, McKinney JS, Berridge MJ, Irvine RF, Putney JW (1984) The second messenger linking receptor activation to internal Ca2+ release in liver. Nature 309:63–66
Cohen ML, Mason N, Wiley KS, Fuller RW (1983) Further evidence that vascular serotonin receptors are of the 5-HT2 type. Biochem Pharmacol 32:567–570
Conn PJ, Sanders-Bush E (1984) Selective 5-HT2 antagonists inhibit serotonin stimulated phosphatidylinositol metabolism in cerebral cortex. Neuropharmacology 8:993–996
Conn PJ, Sanders-Bush E (1985) Serotonin stimulated phosphoinositide turnover: mediation by the S2 binding site in rat cerebral cortex but not in subcortical regions. J Pharmacol Exp Ther 243:195–203
Cortes R, Palacios JM, Pazos A (1984) Visualisation of multiple serotonin receptors in the rat brain by autoradiography. Br J Pharmacol 82:202 P
De Chaffoy de Courcelles D, Leysen JE, De Clerck F, Van Belle H, Janssen PA (1985) Evidence that phospholipid turnover is the signal transducing system coupled to serotonin-S2 receptor sites. J Biol Chem 260:7603–7608
Downes CP, Wusteman MM (1983) Breakdown of polyphosphoinositides and not phosphatidylinositol accounts for muscarinic agonist stimulated inositol phospholipid metabolism in rat parotid glands. Biochem J 216:633–640
Doyle VM, Rüegg UT (1985) Vasopressin induced production of inositol trisphosphate and calcium efflux in a smooth muscle cell line. Biochem Biophys Res Comm 131:469–476
Engel G, Göthert M, Hoyer D, Schlicker E, Hillenbrand K (1986) Identity of inhibitory presynaptic 5-hydroxytryptamine (5-HT) autoreceptors in the rat brain cortex with 5-HT1B binding sites. Naunyn-Schmiedeberg's Arch Pharmacol 332:1–7
Engel G, Göthert M, Müller-Schweinitzer E, Schlicker E, Sistonen L, Stadler PA (1983) Evidence for common pharmacological properties of [3H]-5-hydroxytryptamine autoreceptors in CNS and inhibitory presynaptic 5-hydroxytryptamine receptors on sympathetic nerves. Naunyn-Schmiedeberg's Arch Pharmacol 324:116–124
Engel G, Hoyer D, Kalkman HO, Wick M (1984) Identification of 5-HT2-receptors on longitudinal muscle on the guinea pig ileum. J Rec Res 4:113–126
Fain JM, Berridge MJ (1979) Relationship between hormonal activation of phosphatidylinositol hydrolysis, fluid secretion and calcium flux in the blowfly salivary gland. Biochem J 178:45–58
Gaddum JH, Picarelli ZP (1957) Two kinds of tryptamine receptor. Br J Pharmacol Chem Ther 12:323–328
Hjorth S, Carlsson A, Lindberg P, Sanchez D, Wikström H, Arvidson LE, Hacksell U, Nilsson JLG (1982) 8-Hydroxy-2-(di-N-propylamino)tetralin, 8-OH-DPAT, a potent and selective simplified ergot congener with central 5-HT receptor stimulating activity. J Neural Transm 55:169–188
Hoyer D, Engel G, Kalkman HO (1985a) Characterisation of the 5-HT1B recognition site in rat brain: Binding studies with (−)[125I]iodocyanopindolol. Eur J Pharmacol 118:1–12
Hoyer D, Engel G, Kalkman HO (1985b) Molecular pharmacology of 5-HT1 and 5-HT2 recognition sites in rat and pig brain membranes: Radioligand binding studies with [3H]5-HT, [3H]-OH-DPAT, (−)[125I]cyanopindolol, [3H]mesulergine and [3H]-ketanserin. Eur J Pharmacol 118:13–23
Janowsky A, Labarca R, Paul SM (1984) Characterization of neurotransmitter receptor-mediated phosphatidylinositol hydrolysis in the rat hippocampus. Life Sci 35:1953–1961
Kalkman HO, Boddeke HWGM, Doods HN, Timmermans PBMWM, Van Zwieten PA (1983) Hypotensive activity of serotonin receptor agonists in rats is related to their affinity for 5-HT1 receptors. Eur J Pharmacol 91:155–156
Kaumann AJ, Frenken M (1985) A paradox: the 5-HT2 receptor antagonist ketanserin restores the 5-HT induced contraction depressed by methysergide in large coronary arteries of calf. Naunyn Schmiedeberg's Arch Pharmacol 328:295–300
Kendall DA, Nahorski SR (1985) 5-Hydroxytryptamine stimulated inositol phospholipid hydrolysis in rat cerebral cortex slices: pharmacological characterisation and effects of antidepressants. J Pharmacol Exp Ther 233:473–479
Kimes BW, Brandt BL (1976) Characterisation of two putative smooth muscle cell lines from rat thoracic aorta. Exp Cell Res 98:349–366
Leysen JE, Niemegeers CJE, Van Nueten JM, Laduron PM (1982) [3H]ketanserin (R 41 468), a selective 3H ligand for serotonin 2 receptor binding sites. Mol Pharmacol 21:301–314
Lucki I, Nobler MS, Frazer A (1984) Differential actions of serotonin antagonists on two behavioural models of serotonin receptor activation in the rat. J Pharmacol Exp Ther 228:133–139
Michell RH, Kirk CJ, Jones LM, Downes CP, Creba JA (1981) The stimulation of inositol lipid metabolism that accompanies calcium mobilisation in cells: Defined characteristics and unanswered questions. Philos Trans R Soc Lond 296:123–138
Nabika T, Velletri PA, Lovenberg W, Bevan MA (1985) Increase in cytosolic calcium and phosphoinositide metabolism induced by angiotensin II and [Arg]vasopressin in vascular smooth muscle cells. J Biol Chem 260:4661–4670
Nakaki T, Roth BL, Chuang DM, Costa E (1985) Phasic and tonic components in 5-HT2 receptor-mediated rat aorta contraction: Participation of Ca++ channels and phospholipase C. J Pharmacol Exp Ther 234:442–446
Nishizuka Y (1984) The role of protein kinase C in cell surface signal transduction and tumor promotion. Nature 308:693–698
Pazos A, Hoyer D, Palacios JM (1984a) Mesulergine, a selective serotonin 2 ligand in the rat cortex, does not label these receptors in porcine and human cortex: Evidence for species differences in brain serotonin 2 receptors. Eur J Pharmacol 106:531–538
Pazos A, Hoyer D, Palacios JM (1984b) The binding of serotonergic ligands to the porcine choroid plexus: characterisation of a new type of serotonin recognition site. Eur J Pharmacol 106:539–546
Pedigo NW, Yamamura HI, Nelson DL (1981) Discrimination of multiple [3H]5-hydroxytryptamine binding sites by the neuroleptic spiperone in rat brain. J Neurochem 36:220–226
Peroutka SJ, Snyder SH (1979) Multiple serotonin receptors: differential binding of [3H]5-hydroxytryptamine, [3H]lysergic acid diethylamine and [3H]spiroperidol. Mol Pharmacol 16: 687–699
Peroutka SJ, Lebovitz RM, Snyder SH (1981) Two distinct central serotonin receptors with different physiological functions. Science 212:827–829
Peroutka SJ, Noguchy M, Tolner DJ, Allen GS (1983) Serotonin induced contraction of canine basilar artery mediation by 5-HT1 receptors. Brain Res 259:327–330
Roth BL, Nakaki T, Chuang DM, Costa E (1984) Aortic recognition sites for serotonin (5-HT) are coupled to phospholipase C and modulate phosphatidylinositol turnover. Neuropharmacol 23:1223–1225
Rüegg UT, Doyle VM, Zuber JF, Hof RP (1985) A smooth muscle cell line suitable for the study of voltage sensitive calcium channels. Biochem Biophys Res Commun 130:447–453
Shenker A, Maayani S, Weinstein H, Green JP (1985) Two 5-HT receptors linked to adenylate cyclase in guinea pig hippocampus are described by 5-carboxymido-tryptamine and spiperone. Eur J Pharmacol 109:427–429
Smith JB, Smith L, Brown ER, Barnes D, Sabir MA, Davis JS, Farese RV (1984) Angiotensin II rapidly increases phosphatidate-phosphoinositide synthesis and phosphoinositide hydrolysis and mobilises intracellular calcium in cultured arterial muscle cells. Proc Natl Acad Sci USA 81:7812–7816
Somlyo AV, Bond M, Somlyo AP (1985) Inositol trisphosphate induced calcium release and contraction in vascular smooth muscle. Proc Natl Acad Sci USA 82:5231–5235
Streb H, Irvine RF, Berridge MJ, Schulz I (1983) Release of Ca2+ from a nonmitochondrial intracellular store in pancreatic acinar cells by inositol 1,4,5-trisphosphate. Nature 306:67–69
Suematsu E, Hirata M, Hashimoto T, Kuriyama H (1984) Inositol 1,4,5-trisphosphate releases Ca2+ from intracellular store sites in skinned single cells of porcine coronary artery. Biochem Biophys Res Comm 120:481–485
Yano K, Higshida H, Hattori H, Nozawa Y (1985) Bradykinininduced transient accumulation of inositol trisphosphate in neuron-like cell line NG 108-15 cells. FEBS Lett 181:403–406
Author information
Authors and Affiliations
Additional information
Part of this work has been presented in London, December 1985, at the winter meeting of the British Pharmacological Society
Rights and permissions
About this article
Cite this article
Doyle, V.M., Creba, J.A., Rüegg, U.T. et al. Serotonin increases the production of inositol phosphates and mobilises calcium via the 5-HT2 receptor in A7r5 smooth muscle cells. Naunyn-Schmiedeberg's Arch. Pharmacol. 333, 98–103 (1986). https://doi.org/10.1007/BF00506510
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00506510