Summary
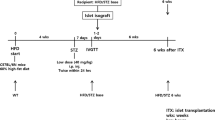
The effect of donor islet mass on the metabolic normalization in streptozotocin-diabetic rats was examined by comparing normal control rats with two transplant groups receiving 800 or 2,000 islets intraportally, i.e. grafts containing respectively, 0.8 or 2 million Beta cells at 70% purity. After transplantation, basal glycaemia and daily urine volumes normalized in all recipients and remained normal in both groups over the 20-week follow-up period. After intragastric glucose challenge, the 800-islet group exceeded the normal range of glycaemia at all tested time points (0–120 min) whereas the 2,000-islet group exhibited higher glycaemia only at 15 and 30 min; no deterioration in glucose tolerance occurred during the 20-week follow-up. Both transplant groups presented lower serum fructosamine levels than diabetic control rats, normal levels being maintained only in the 2000-islet group. At post-transplantation week 21, both recipient groups exhibited the same body weight as age-matched normal control rats, but their serum triglyceride levels were significantly higher. Hepatic insulin contents were comparable to the insulin contents of the grafts at the time of implantation, representing 16 or 40% of the pancreatic insulin content in the age-matched control rats. Although islet grafts with 0.8 million Beta cells can restore basal glycaemia in adult streptozotocin-diabetic rats, implants of 2 million Beta cells are necessary to correct other parameters of glucose homeostasis. The present data indicate the need of determining the number of grafted Beta cells in studies on the metabolic outcome of islet cell transplantation. DNA content and percentage of Beta cells in the graft are proposed as useful parameters.
Article PDF
Similar content being viewed by others
References
Kemp CB, Knight MJ, Scharp DW, Lacy PE, Ballinger WF (1973) Transplantation of isolated pancreatic islets into the portal vein of diabetic rats. Nature 244: 447 (Letter)
Warnock GL, Rajotte RV (1988) Critical mass of purified islets that induce normoglycemia after implantation into dogs. Diabetes 37: 467–470
Sutton R, Gray DW, McShane P, Peters M, Morris PJ (1987) The metabolic efficiency and long-term fate of intra-portal islet grafts in the cynomolgus monkey. Transplant Proc 19: 3575–3576
Warnock GL, Kneteman NM, Ryan E, Seelis REA, Rabinovitch A, Rajotte RV (1991) Normoglycaemia after transplantation of freshly isolated and cryopreserved pancreatic islets in type 1 (insulin-dependent) diabetes mellitus. Diabetologia 34: 55–58
Orloff MJ, Macedo A, Greenleaf GE, Girard B (1988) Comparison of the metabolic control of diabetes achieved by whole pancreas transplantation and pancreatic islet transplantation in rats. Transplantation 45: 307–312
Hiller WF, Klempnauer J, Luck R, Steiniger B (1991) Progressive deterioration of endocrine function after intraportal but not kidney subcapsular rat islet transplantation. Diabetes 40: 134–140
Alejandro R, Cutfield RG, Shienvold FL et al. (1986) Natural history of intrahepatic canine islet cell autografts. J Clin Invest 78: 1339–1348
Kaufman DB, Morel P, Field MJ, Munn SR, Sutherland DE (1990) Purified canine islet autografts. Transplantation 50: 385–391
Pipeleers DG, Pipeleers-Marichal MA, Karl IE, Kipnis DM (1978) Secretory capability of islets transplanted intraportally in the diabetic rat. Diabetes 27: 817–824
Trimble ER, Siegel ER, Berthoud HR, Renold AE (1980) Intraportal islet transplantation: functional assessment in conscious unrestrained rats. Endocrinology 106: 791–797
Pipeleers DG, Pipeleers-Marichal M, Hannaert JC et al. (1991) Transplantation of purified islet cells in diabetic rats I. Standardization of islet cell grafts. Diabetes 40: 908–919
Pipeleers-Marichal MA, Pipeleers DG, Cutler J, Lacy PE, Kipnis DM (1976) Metabolic and morphologic studies in intraportal-islet-transplanted rats. Diabetes 25: 1041–1051
Baker J, Metcalf P, Scragg R, Johnson R (1991) Fructosamine Test-Plus, a modified fructosamine assay evaluated. Clin Chem 37: 552–556
Bartels H, Bohmer M (1971) Eine Micromethode zur Kreatininbestimmung. Clin Chim Acta 32: 81–85
Wahlefeld AW (1974) Triglycerides. Determination after enzymatic hydrolysis. In: Bergmeyer HU (ed) Methods of enzymatic analysis. Verlag Chemie, Weinheim and Academic Press, New York, London pp 1831–1839
Siedel J, Hagele EO, Ziegenhorn J, Wahlefeld AW (1983) Reagent for the enzymatic determination of serum total cholesterol with improved lipolytic efficiency. Clin Chem 29: 1075–1080
Pipeleers DG, Pipeleers-Marichal M, Vanbrabandt B, Duys S (1991) Transplantation of purified islet cells in diabetic rats II. Immunogenicity of allografted islet β-cells. Diabetes 40: 920–930
Pipeleers DG, In't Veld PA, Van De Winkel M, Maes E, Schuit FC, Gepts W (1985) A new in vitro model for the study of pancreatic A and B cells. Endocrinology 117: 806–816
Pipeleers D (1987) The biosociology of pancreatic B cells. Diabetologia 30: 277–291
Orci L (1982) Macro and micro-domains in the endocrine pancreas. Diabetes 31: 538–565
Bonner-Weir S, Orci L (1982) New perspectives on the microvasculature of the islets of Langerhans in the rat. Diabetes 31: 883–889
Vialettes B, Sutherland DER, Matas AJ, Payne WD, Najarian JS (1979) Amelioration of streptozotocin-induced diabetes in rats: effect of islet isografts on plasma lipids and other metabolic abnormalities. Metabolism 28: 489–494
Hesse U, Sutherland DER, Florack G, Field J, Ward S, Najarian JS (1986) Comparison of two methods of islet preparation and transplantation in dogs. Diabetes 35: 1109–1118
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Keymeulen, B., Teng, H., Vetri, M. et al. Effect of donor islet mass on metabolic normalization in streptozotocin-diabetic rats. Diabetologia 35, 719–724 (1992). https://doi.org/10.1007/BF00429090
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF00429090