Abstract
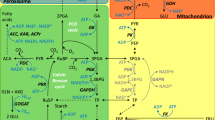
A mutant line, RPr79/2, of barley (Hordeum vulgare L. cv. Maris Mink) has been isolated that has an apparent defect in photorespiratory nitrogen metabolism. The metabolism of 14C-labelled glutamine, glutamate and 2-oxoglutarate indicates that the mutant has a greatly reduced ability to synthesise glutamate, especially in air, although in-vitro enzyme analysis indicates the presence of wild-type activities of glutamine synthetase (EC 6.3.1.2) glutamate synthase (EC 1.4.7.1 and EC 1.4.1.14) and glutamate dehydrogenase (EC 1.4.1.2). Several characteristics of RPr79/2 are very similar to those described for glutamate-synthase-deficient barley and Arabidopsis thaliana mutants, including the pattern of labelling following fixation of 14CO2, and the rapid rise in glutamine content and fall in glutamate in leaves on transfer to air. The CO2-fixation rate in RPr79/2 declines much more slowly on transfer from 1% O2 to air than do the rates in glutamate-synthase-deficient plants, and RPr79/2 plants do not die in air unless the temperature and irradiance are high. Analysis of (glutamine+NH3+2-oxoglutarate)-dependent O2 evolution by isolated chloroplasts shows that chloroplasts from RPr79/2 require a fivefold greater concentration of 2-oxoglutarate than does the wild-type for maximum activity. The levels of 2-oxoglutarate in illuminated leaves of RPr79/2 in air are sevenfold higher than in Maris Mink. It is suggested that RPr79/2 is defective in chloroplast dicarboxylate transport.
Similar content being viewed by others
References
Anderson, J.W., Done, J. (1977a) A polarographic study of glutamate synthase activity in isolated chloroplasts. Plant Physiol. 60, 354–359
Anderson, J.W., Done, J. (1977b) Polarographic study of ammonia assimilation by isolated chloroplasts. Plant Physiol. 60, 504–508
Bright, S.W.J., Lea, P.J., Arruda, P., Hall, N.P., Kendall, A.C., Keys, A.J., Kueh, J.S.H., Parker, M.L., Rognes, S.E., Turner, J.C., Wallsgrove, R.M., Miflin, B.J. (1984) Manipulation of the pathways of photorespiration and amino acid metabolism by mutation and selection. In: The genetic manipulation of plants and its application to agriculture, pp. 141–169, Lea, P.J., Stewart, G.P., eds. Oxford University Press, Oxford
Dry, I.B., Wiskich, J.T. (1983) Characterization of dicarboxylate stimulation of ammonia, glutamine, and 2-oxoglutarate-dependent O2 evolution in isolated pea chloroplasts. Plant Physiol. 72, 291–296
Kendall, A.C., Wallsgrove, R.M., Hall, N.P., Turner, J.C., Lea, P.J. (1986) Carbon and nitrogen metabolism in barley (Hordeum vulgare L.) mutants lacking ferredoxin-dependent glutamate synthase. Planta 168, 316–323
Keys, A.J., Bird, I.F., Cornelius, M.J., Lea, P.J., Wallsgrove, R.M., Miflin, B.J., (1978) The photorespiratory nitrogen cycle. Nature 275, 741–743
Larsen, R.O., Cornwall, K.L., Gee, S.L., Bassham, J.A. (1981) Amino acid synthesis in photosynthesising spinach cells: effect of ammonia on pool sizes and rates of labelling from [14C]O2. Plant Physiol. 68, 292–299
Lilley, R.M., Fitzgerald, M.P., Rientis, K.G., Walker, D.A. (1975) Criteria of intactness and the photosynthetic activity of spinach chloroplast preparations. New Phytol. 75, 1–10
Mann, A.F., Fentem, P.A., Stewart, G.R. (1980) Tissue localization of barley (Hordeum vulgare L.) glutamine synthetase isozymes. FEBS Lett. 110, 265–267
Somerville, C.R. (1984) The analysis of photosynthetic carbon dioxide fixation and photorespiration by mutant selection. In: Oxford Surveys of Plant Molecular and Cell Biology, vol. 1, pp. 103–131, Miflin, B.J., ed. Oxford University Press, Oxford
Somerville, C.R., Ogren, W.L. (1980) Inhibition of photosynthesis in Arabidopsis mutants lacking in leaf glutamate synthase activity. Nature 286, 257–259
Somerville, S.C., Ogren, W.L. (1983) An Arabidopsis thaliana mutant defective in chloroplast dicarboxylate transport. Proc. Natl. Acad. Sci. USA 80, 1290–1294
Walker, K.A., Givan, C.V., Keys, A.J. (1984) Glutamic acid metabolism and the photorespiratory nitrogen cycle in wheat leaves: metabolic consequences of elevated ammonia concentrations and of blocking ammonia assimilation. Plant Physiol. 75, 60–65
Wallsgrove, R.M., Harel, E., Lea, P.J., Miflin, B.J. (1977) Studies on glutamate synthase from the leaves of higher plants. J. Exp. Bot. 28, 588–596
Wallsgrove, R.M., Lea, P.J., Miflin, B.J. (1983) Intracellular localization of aspartate kinase and the enzymes of threonine and methionine biosynthesis in green leaves. Plant Physiol. 71, 780–784
Wallsgrove, R.M., Lea, P.J., Miflin, B.J. (1979) Distribution of the enzymes of nitrogen assimilation within the pea leaf cell. Plant Physiol. 63, 232–236
Wintermans, J.F.G.M., Demots, A. (1965) Spectrophotometric characteristics of chlorophylls a and b and their pheophytins in ethanol. Biochim. Biophys. Acta 109, 448–453
Woo, K.C., Osmond, C.B. (1983) Characterization of dicarboxylate stimulation of ammonia, glutamine and 2-oxoglutarate-dependent O2 evolution in isolated pea chloroplasts. Plant Physiol. 72, 291–296
Williamson, J.R., Corkey, B.E. (1979) Assay of citric acid cycle intermediates and related compounds — update with tissue metabolite levels and intracellular distribution. Methods Enzymol. 55, 200–222
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Wallsgrove, R.M., Kendall, A.C., Hall, N.P. et al. Carbon and nitrogen metabolism in a barley (Hordeum vulgare L.) mutant with impaired chloroplast dicarboxylate transport. Planta 168, 324–329 (1986). https://doi.org/10.1007/BF00392356
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00392356