Abstract
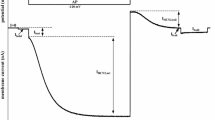
Inactivation of Ca2+ -induced Cl− currents was studied in Xenopus oocytes using the two-electrode voltage-clamp technique. In oocytes permeabilized to Ca2+ by treatment with the ionophore A23187, Ca2+ influx caused by the addition of 2.5–5 mM Ca2+ to the extracellular solution elicited Cl− currents consisting of two components: a fast, transient one (I fast) and a slow one (I slow). In response to a subsequent application of the same dose of Ca2+, I fast and I slow were reduced (inactivation phenomenon). The inactivation did not depend on the direction of current flow, but did depend on the duration of the first exposure to Ca2+. The extent of inactivation of I fast was more significant than that to I slow. Both I fast and I slow fully recovered from inactivation in less than 30 min. Intracellular injections of 100–400 pmol CaCl2 evoked large inward currents but did not reduce the amplitude of currents evoked by Ca2+ influx. The activator of protein kinase C, β-phorbol dibutyrate, caused full inhibition of I fast without any change in I slow. H-7 (1,5-isoquinolinesulfonyl-1,2 methylpiperazine), an inhibitor of protein kinases, strongly reduced the extent of inactivation. Our results suggest that elevation of intracellular Ca2+ by Ca2+ influx through the plasma membrane causes inactivation of the Ca2+ -dependent Cl− conductance via activation of a Ca2+ -dependent protein kinase, possibly protein kinase C, whereas Ca2+ arriving at the membrane from inside the cell does not initiate the processes leading to inactivation.
Similar content being viewed by others
References
Alkon DL, Rasmussen H (1988) A spatial-temporal model of cell activation. Science 239: 998–1005
Barish ME (1983) A transient calcium-dependent chloride current in the immature Xenopus oocyte. J Physiol (Lond) 342: 309–325
Boton R, Gillo B, Dascal N, Lass Y (1989) Two calcium-activated chloride conductance in Xenopus laevis oocytes permeabilized with the ionophore A23187. J Physiol (Lond) 408: 511–534
Dascal N (1987) The use of Xenopus oocytes for the study of ion channels. CRC Crit Rev Biochem 22: 317–387
Dascal N, Landau EM, Lass Y (1984) Xenopus oocyte resting potential, muscarinic responses and the role of calcium and cyclic GMP. J Physiol (Lond) 352: 551–574
Dascal N, Gillo B, Lass Y (1985) Role of calcium mobilization in mediation of acetylcholine-evoked chloride current in Xenopus laevis oocytes. J Physiol (Lond) 366: 299–314
Dascal N, Ifune C, Hopkins R, Snutch TP, Lubbert H, Simon MI, Davidson N, Lester HA (1986a) Involvement of a GTP-binding protein in mediation of serotonin and acetylcholine responses in Xenopus oocytes injected with rat brain messenger RNA. Mol Brain Res 1: 201–209
Dascal N, Snutch TP, Lubbert H, Davidson N, Lester HA (1986b) Expression and modulation of voltage operated calcium channels after RNA injection in Xenopus oocytes. Science 231: 1147–1150
Dumont JN (1972) Oogenesis in Xenopus laevis (Daudin). 1. Stages of oocyte development in laboratory maintained animals. J Morphol 136: 153–180
Evans MG, Marty A (1986) Calcium-dependent chloride current in isolated cells from rat lacrimal gland. J Physiol (Lond) 378: 437–460
Garland LG, Bonser RW, Thompson NT (1987) Protein kinase C inhibitors are not selective. Trends Pharmacol Sci 8: 33
Kato K-I, Kaneko S, Nomura Y (1988) Phorbol ester inhibition of current response and simultaneous protein phosphorylation in Xenopus oocytes injected with brain mRNA. J Neurochem 50: 766–773.
Lester HA (1988) Heterologous expression of excitability proteins: route to more specific drugs? Science 241: 1057–1063
Levitan IB (1985) Phosphorylation of ion channels. J Membr Biol 87: 177–190
Levitan IB (1988) Modulation of ion channels in neurons and other cells. Annu Rev Neurosci 11: 119–136
Lupu-Meiri M, Shapira H, Oron Y (1988) Hemispheric asymmetry of rapid chloride responses to inositol trisphosphate and calcium in Xenopus oocytes. FEBS Lett 240: 83–87
Lupu-Meiri M, Shapira H, Oron Y (1988) Dual regulation by protein kinase C of the muscarinic response in Xenopus oocytes. Pflügers Arch 413: 498–504
Marty A, Tan YP, Trautmann A (1984) Three types of calcium-dependent channels in rat lacrimal gland. J Physiol (Lond) 357: 293–325
Miledi R (1982) A calcium-dependent transient outward current in Xenopus laevis oocytes. Proc R Soc Lond [Biol] 215: 492–497
Miledi R, Parker I (1984) Chloride current induced by injection of calcium into Xenopus oocytes. J Physiol (Lond) 357: 173–183
Moran O, Dascal N (1989) Protein kinase C modulates neurotransmitter responses in Xenopus oocytes injected with rat brain RNA. Mol Brain Res 5: 193–202
Nishizuka Y (1984) The role of protein kinase C in cell surface signal transduction and tumor promotion. Nature 308: 693–698
Nishizuka Y (1986) Studies and perspectives of protein kinase C. Science 233: 305–312
Nishizuka Y (1988) The molecular heterogeneity of protein kinase C and its implications for cellular regulation. Nature 334: 661–665
Singer D, Boton R, Moran O, Dascal N (1990) Short-and long-lasting desensitization of serotonergic response in Xenopus oocytes injected with rat brain RNA: roles for inositol 1,4,5-trisphosphate and protein kinase C. Pflügers Arch 416: 7–16
Takahashi T, Neher E, Sakmann B (1987). Rat brain serotonin receptors in Xenopus oocytes are coupled by intracellular calcium to endogenous channels. Proc Natl Acad Sci USA 84: 5063–5067
Taleb O, Feltz P, Bossu J-L, Feltz A (1988) Small-conductance chloride channels activated by calcium on cultured endocrine cells from mammalian pars intermedia. Pflügers Arch 412: 641–646
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Boton, R., Singer, D. & Dascal, N. Inactivation of calcium-activated chloride conductance in Xenopus oocytes: roles of calcium and protein kinase C. Pflügers Arch 416, 1–6 (1990). https://doi.org/10.1007/BF00370214
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00370214