Abstract
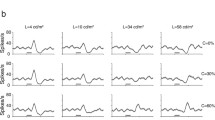
Neurons in area 17 of the cat visual cortex adapt when stimulated by drifting patterns of optimal orientation, spatial frequency and temporal frequency (Ohzawa et al. 1982; Albrecht et al. 1984; Ohzawa et al. 1985). A component of this adaptation has been attributed to a contrast gain-control mechanism, rather than to neural fatigue, and results in enhanced differential sensitivity around the adapting contrast level (Ohzawa et al. 1982; Albrecht et al. 1984; Ohzawa et al. 1985). Experiments described here suggest that neural response rate, the directional selectivity of the cell, and the temporal frequency of the stimulus, are the principal determinants of adaptation, irrespective of other stimulus parameters such as contrast, velocity, or spatial frequency. The present results can nevertheless accommodate the results of previous studies of adaptation, and additionally provide scope for the resolution of apparent contradictions between results from psychophysical and neurophysiological studies of adaptation.
Similar content being viewed by others
References
Albrecht DG, Hamilton DB (1982) Striate cortex of monkey and cat: contrast response function. J Neurophysiol 48:217–237
Albrecht DG, Farrer SB, Hamilton DB (1984) Spatial contrast adaptation characteristics of neurons recorded in the cat's visual cortex. J Physiol 347:713–739
Barlow HB, Brindley GS (1963) Inter-ocular transfer of movement after-effects during pressure blinding of the stimulated eye. Nature 200:1347
Barlow HB, Macleod DIA, van Meeteren A (1976) Adaptation to gratings: no compensatory advantages found. Vision Res 16:1043–1045
Blakemore C, Campbell FW (1969) On the existence in the human visual system of neurons selectively sensitive to the orientation and size of retinal images. J Physiol (London) 203:237–260
Borst A, Egelhaaf M (1987) Temporal modulation of luminance adapts time constant of fly movement detectors. Biol Cybern 56:209–215
Bullier J, Henry GH (1979) Laminar distribution of first-order neurons and afferent terminals in cat striate cortex. J Neurophysiol 42:1251–1263
Cleland BG, Dubin MW, Levick WR (1971a) Sustained and transient neurone in the cat's retina and lateral geniculate nucleus. J Physiol (London) 217:473–497
Cleland BG, Dubin MW, Levick WR (1971b) Simultaneous recording of input and output of lateral geniculate neurone. Nat New Biol 231:191–192
Dobson AJ (1983) An introduction to statistical modelling. Chapman & Hall, London
Gilinsky AS (1968) Orientation-specific effects of adapting light on visual acuity. J Opt Soc Am 58:13–18
Hammond P (1978) Inadequacy of nitrous oxide/oxygen mixtures for maintaining anaesthesia in cats: satisfactory alternatives. Pain 5:142–151
Henry GH (1977) Receptive field classes of cells in the striate cortex of the cat. Brain Res 133:1–28
Hubel DH, Wiesel TN (1962) Receptive fields, binocular interaction and functional architecture in the cat's visual cortex. J Physiol (London) 160:106–154
Johnston A, Wright MJ (1983) Visual motion and cortical velocity. Nature 304:436–438
Kaplan E, Shapley RM (1984) The origin of theS(slow) potential in the mammalian lateral geniculate nucleus. Exp Brain Res 55:111–116
Lorenceau J (1987) Recovery from contrast adaptation: effects of spatial and temporal frequency. Vision Res 27:2185–2191
McCullagh P, Nelder JA (1983) Generalized linear models. Chapman & Hall, London
McKee SP (1981) A local mechanism for differential velocity detection. Vision Res 21:491–500
MacKerras P, Bossomaier T, Maddess T, Laughlin S (1988) Information gains from contrast adaptation (in preparation)
Maddess T (1986) Afterimage-like effects in the motion-sensitive neuronH1. Proc R Soc London B 228:433–459
Maddess T, Laughlin SB (1985) Adaptation of the motionsensitive neuronH1 is generated locally and governed by contrast frequency. Proc R Soc London B 225:251–275
Movshon JA, Lennie P (1979) Pattern-selective adaptation in visual cortical neurons. Nature 278:850–852
Nakayama K (1981) Differential motion hyperacuity under conditions of common image motion. Vision Res 21:1475–1482
Ohzawa I, Sclar G, Freeman RD (1982) Contrast gain control in the cat visual cortex. Nature 298:266–268
Ohzawa I, Sclar G, Freeman RD (1985) Contrast gain control in the cat visual system. J Neurophysiol 54:651–667
Pantle A (1974) Motion aftereffect magnitude as a measure of the spatio-temporal response properties of direction-sensitive analyzers. Vision Res 14:1229–1236
Pantle A, Sekuler RW (1968) Size-detecting mechanisms in human vision. Science 162:1146–1148
Rodieck RW, Pettigrew JD, Bishop PO, Nikara T (1967) Residual eye movements in receptive field studies of paralyzed cats. Vision Res 7:107–110
Santen JPH van, Sperling G (1985) Elaborated Reichardt detectors. J Opt Soc Am A 2:300–321
Sclar G (1987) Expression of “retinal” contrast gain control by neurons of the cat's lateral geniculate nucleus. Exp Brain Res 66:589–596
Sekuler R (1975) Visual motion perception. In: Carterette EC, Freeman MP (eds) Seeing. Handbook of perception, vol V. Academic Press, New York, pp 387–430
Sekuler R, Pantle A, Levinson E (1978) Physiological basis of motion perception. In: Held R, Leibowitz HW, Teuber H (eds) Perception. Handbook of sensory physiology, vol VIII Springer, Berlin Heidelberg New York, pp 67–98
Shapley RM, Victor JD (1978) The effect of contrast on the transfer properties of cat retinal ganglion cells. J Physiol London 285:275–298
Tohlhurst DJ, Movshon JA (1975) Spatial and temporal contrast sensitivity of striate cortical neurons. Nature 257:674–675
Van Doorn AJ, Koenderink JJ (1983) Detectability of velocity gradients in moving random dot patters. Vision Res 23:799–804
Weisberg S (1980) Applied linear regression. Wiley, New York
Wright MJ, Johnston A (1985) Invariant tuning of motion aftereffect. Vision Res 25:1947–1955
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Maddess, T., McCourt, M.E., Blakeslee, B. et al. Factors governing the adaptation of cells in area-17 of the cat visual cortex. Biol. Cybern. 59, 229–236 (1988). https://doi.org/10.1007/BF00332911
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/BF00332911