Summary
The initial steps of neuralization by denatured embryonic tissue were studied in single and double layer Triturus ectoderm explants.
-
1.
In single layer explants only part of the responding ectoderm is neuralized. A vesicle with continuous epidermoid envelope develops, from which neuralized spindle-shaped cell groups retract into the interior. Several neural cell groups fuse with each other and form a cushion of neural tissue between implant and vesicle wall. Neural tissue thus never comes into contact with the culture medium but is always protected by an epidermoid envelope.
-
2.
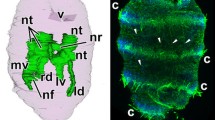
In double layer explants the inner layer is completely neuralized. No epidermoid tissue remains. The inhibitory effect of single layer ectoderm is unblocked by the second envelope, which now furnishes the epidermoid protective vesicle. A thick layer of neural tissue encloses and faces the dead implant. The retinal character of this tissue soon becomes apparent by elimination of micropigment away from the implant (transparence).
-
3.
Three to four days after the operation a process of self-organization begins in this thick layer of retinal tissue. It splits parallel to the surface and forms several retinal vesicles. The inner vesicle walls continue histo-and morphodifferentiation in the original direction, while in the outer walls this direction is reversed: micropigment now is eliminated toward the ventricle-like cavity and retina fronts (eyecups) face the outer epidermoid envelope.
-
4.
The discussion deals with the inhibition of total neuralization in single layer explants and its unblocking by a second envelope, and with the process of splitting and self-organization of induced retinal tissue. Retina induction in explants is compared with normal and artificial neural and retina induction in embryos.
Similar content being viewed by others
References
Berrill NJ, Karp G (1976) Development, Mc Graw-Hill, New York
Burnside B (1971) Microtubules and microfilaments in newt neurulation. Dev Biol 26:434
Grobstein C (1955) Tissue disaggregation in relation to determination and stability of cell type. Ann NY Acad Sci 60: 1095–1106
Harrison RG (1969) Organization and development of the embryo. Yale Univ Press, New Haven and London
Holtfreter J (1943) A Study of the mechanics of gastrulation, Part I. J Exp Zool 94:261–318
Jacobson CD (1962) Cell migration and the process of neurulation in the axolotl larva. Zool Bidrag Uppsala 35:433–449
Kohonen J, Paranko J (1980) Cell movement and ultrastructure of isolated amphibian gastrula ectoderm: A scanning and transmission electron microscopical study. Second International Congress on Cell Biology Berlin
Lopashov GV (1935) Die Entwicklungsleistungen des Gastrulaektoderms in Abhängigkeit von Veränderungen der Masse. Biol Zbl 58:606–615
Rollhäuser-ter Horst J (1953) Die Induktionsleistung verschiedener Tritonkeimteile nach Denaturierung. W Roux' Arch 146: 183–200
Rollhäuser-ter Horst J (1977) Artificial neural induction in amphibia. I. Sandwich explants. II. Host embryos. Anat Embryol 151: 309–324
Rollhäuser-ter Horst J (1979) Artificial neural crest formation in amphibia. Anat Embryol 157:113–120
Saxén L (1977) Directive versus permissive induction: A working hypothesis. In: Lash JW, Burger MM (ed) Cell and Tissue Interactions. Raven Press, New York
Saxén L, Toivonen S (1962) Primary embryonic induction, London: Academic Press Inc
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Horst, J.Rt. Artificial neural induction in amphibia. Anat Embryol 162, 69–80 (1981). https://doi.org/10.1007/BF00318095
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00318095