Abstract
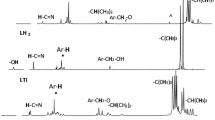
The polymerization of ε-caprolactone (CL) using triphenyltin acetate has been investigated. Polymers with broad MWD were obtained in almost quantitative yield. After an initial induction period, the polymerization showed zero order kinetics with respect to monomer and nearly first order with respect to catalyst. The mechanism of the propagation reaction is proposed to proceed via an ester alcoholysis, with initiation by traces of water. The zero order kinetics are explained as follows: the tin catalyst is coordinated by the cyclic lactone, monomer addition to the polymer chain in the rate-determining step results in formation of an acyclic ester link, which is no longer a strong donor, and leads to a coordinatively unsaturated tin species.
Similar content being viewed by others
References
I. Goodman in Encyclopedia of Polymer Science and Technology, 2nd Ed., vol. 12, p. 36 (1988).
A. Schindler, Y.M. Hibionada, C.G. Pitt, J. Polymer Sci., Polymer Chem. Ed., 1982, 20, 319.
J.W. Leenslag and A.J. Rennings, Makromol. Chem., 1987, 188, 1809.
H.R. Kricheldorf, M. Berl, N. Scharnagl, Macromolecules, 1988, 21, 286.
H.R. Kricheldord and M. Sumbel, Eur. Polymer. J., 1989, 25, 585.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Bassi, M.B., Padias, A.B. & Hall, H.K. The hydrolytic polymerization of ε-caprolactone by triphenyltin acetate. Polymer Bulletin 24, 227–232 (1990). https://doi.org/10.1007/BF00297322
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00297322