Summary
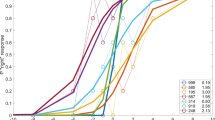
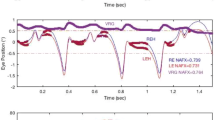
Ocular vergence movements were measured with a scleral coil technique under stabilized viewing conditions for disparity. Crossed disparity steps, ranging between 0.25 and 10 deg, of three different targets were imposed. Ocular vergence responses consisted of converging movements with an initially constant velocity. This velocity increased with the magnitude of disparity up to about 4 deg and decreased for larger disparities. For disparities up to 2 deg the responses saturated at the limit of convergence. For larger disparities responses were transient, i.e. after large converging movements the angle of convergence gradually declined to about its initial value. For disparities larger than 5 deg amplitudes of the transient responses decreased and occasionally responses were completely absent. The transient character of responses was apparently due to adaptation of the vergence system to a specific disparity, since responses to different disparities could still be induced. Probing of the vergence system with two successive disparity steps of different magnitudes showed that adaptation was selective for a limited range of disparities around the adapting disparity stimulus. Stabilized disparity pulses with durations ranging from 100 to 800 ms induced ocular vergence movements following the time integral of disparity rather than momentary disparity. This indicated that the part of the vergence system sensitive to disparity has mainly integrative properties for large as well as small disparities. Stimulation with trains of shortlasting disparity pulses showed that adaptation also occurred under normal viewing conditions. They further revealed that adaptation is erased during a short period after each blink.
Similar content being viewed by others
References
Barlow HB, Blakemore C, Pettigrew JD (1967) The neural mechanisms of binocular depth discrimination. J Physiol 193: 327–342
Cumming BG, Judge SJ (1986) Disparity-induced and blur-induced convergence eye movement and accomodation in the monkey during dichoptic viewing of moving random-dot stereograms. J Neurophysiol 55: 896–914
Collewijn H, Van der Mark F, Jansen TC (1975) Precise recording of human eye movements. Vision Res 15: 447–450
Erkelens CJ, Collewijn H (1985a) Eye movements in relation to loss and regaining of fusion of disjunctively moving random-dot stereograms. Human Neurobiol 4: 181–188
Erkelens CJ, Collewijn H (1985b) Eye movements and stereopsis during dichoptic viewing of moving random-dot stereograms. Vision Res 25: 1689–1700
Erkelens CJ, Collewijn H (1985c) Motion perception during dichoptic viewing of moving random-dot stereograms. Vision Res 25: 583–588
Erkelens CJ, Regan D (1986) Human ocular vergence movements induced by changing size and disparity. J Physiol 379: 145–169
Fender D, Julesz B (1967) Extension of Panum's fusional area in binocularly stabilized vision. JOSA 57: 819–830
Hyson MT, Julesz B, Fender D (1983) Eye movements and neural remapping during fusion of misaligned random-dot stereograms. JOSA 73: 1665–1673
Jones R, Kerr KW (1971) Motor responses to conflicting asymmetrical vergence stimulus information. Am J Optom 48: 989–1000
Jones R (1980) Fusional vergence: sustained and transient components. Am J Optom Physiol Opt 57: 640–644
Julesz B (1978) Global stereopsis: cooperative phenomena in stereoscopic depth perception. In: Held R, Leibowitz HW, Teuber H-L (eds) Handbook of sensory physiology. Vol VIII. Springer, Berlin, pp 215–264
Krishnan VV, Stark L (1977) A heuristic model for human vergence eye movement system. IEEE Trans Biomed Electr BME-24: 44–49
Maddox EE (1893) The clinical use of prisms, 2nd ed. John Wright Sons, Bristol
Marr D (1982) Stereopsis. In: Vision, Chap 3.3. WH Freeman, San Francisco
Mitchell DE (1970) Properties of stimuli eliciting vergence eye movements and stereopsis. Vision Res 10: 145–162
Poggio GF, Fischer B (1977) Binocular interaction and depth sensitivity in striate and prestriate cortex of behaving rhesus monkey. J Neurophysiol 40: 1392–1405
Poggio GF, Poggio T (1984) The analysis of stereopsis. Ann Rev Neurosci 7: 379–412
Rashbass C, Westheimer G (1961) Disjunctive eye movements. J Physiol 159: 339–360
Rashbass C (1981) Reflexions on the control of vergence. In: Zuber BL (ed) Models of oculomotor behavior and control. CRC Press, Boca Raton, pp 139–148
Regan D, Erkelens CJ, Collewijn H (1986) Necessary conditions for motion in depth perception. Inv Ophthalmol Vis Sci 27: 584–597
Richards W (1971) Anomalous stereoscopic depth perception. JOSA 61: 410–414
Riggs LA, Niehl EW (1960) Eye movements recorded during convergence and divergence. JOSA 50: 913–920
Robinson DA (1963) A method of measuring eye movement using a scleral coil in a magnetic field. IEEE Trans Biomed Electr BME 10: 137–145
Semmlow JL, Hung GK, Ciuffreda KJ (1986) Quantitative assessment of disparity vergence components. Inv Ophthalmol Vis Sci 27: 558–564
Westheimer G (1979) Cooperative neural processes involved in stereoscopic acuity. Exp Brain Res 36: 585–597
Westheimer G, Mitchell DE (1956) Eye movement responses to convergence stimuli. Arch Ophthalmol 55: 848–856
Westheimer G, Mitchell DE (1969) The sensory stimulus for disjunctive eye movements. Vision Res 9: 749–755
Zuber BL, Stark L (1968) Dynamic characteristics of the fusional vergence eye-movement system. IEEE Trans Syst Sci Cybern SSC-4: 72–79
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Erkelens, C.J. Adaptation of ocular vergence to stimulation with large disparities. Exp Brain Res 66, 507–516 (1987). https://doi.org/10.1007/BF00270683
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00270683