Abstract
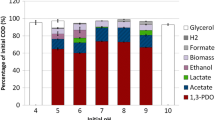
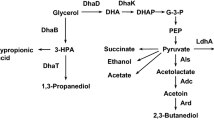
The conversion of glycerol to 1,3-propanediol by Citrobacter freundii DSM 30040 was optimized in single- and two-stage continuous cultures. The productivity of 1,3-propanediol formation was highest under glycerol limitation and increased with the dilution rate (D) to a maximum of 3.7 g·l−1·h−1. Glycerol dehydratase seemed to be the rate-limiting step in 1,3-propanediol formation. Conditions for the two-stage fermentation process were as follows: first stage, glycerol limitation (250mM), pH 7.2, D=0.1 h−, 31° C; second stage, additional glycerol, pH 6.6, D=0.05 h−1, 28° C. Under these conditions 875mM glycerol were consumed, the final 1,3-propanediol concentration was 545mM, and the overall productivity 1.38 g·1−1·h−1.
Similar content being viewed by others
References
Abeles RH, Brownstein AM, Randles CH (1960) β-Hydroxypropionaldehyde, an intermediate in the formation of 1,3-propanediol by Aerobacter aerogenes. Biochim Biophys Acta 41:530–531
Biebel H (1991) Glycerol fermentation of 1,3-propanediol by Clostridium butyricum. Measurement of product inhibition by use of a pH-auxostat. Appl Microbiol Biotechnol 35:701–705
Bradford MM (1976) A rapid and sensitive method for the quantification of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254
Eggstein M, Kulmann E (1974) Triglycerides and glycerol determination after alkaline hydrolysis. In: Bergmeyer HU (ed) Methoden der enzymatischen Analysen. 3rd edn. Verlag Chemie, Weinheim, pp 1871–1878
Dabrock B, Bahl H, Gottschalk G (1992) Parameters affecting solvent production by Clostridium pasteurianum. Appl Environ Microbiol 58:1233–1239
Forsberg C (1987) Production of 1,3-propanediol from glycerol by Clostridium acetobutylicum and other Clostridium species. Appl Environ Microbiol 53:639–643
Homann T, Tag C, Biebl H, Deckwer WD, Schink B (1990) Fermentation of glycerol to 1,3-propanediol by Klebsiella and Citrobacter strains. Appl Microbiol Biotechnol 33:121–126
Johnson EA, Lin ECC (1987) Klebsiella pneumoniae 1,3-propanediol: NAD+ oxidoreductase. J Bacteriol 169:2050–2054
La Riviere JWM (1958) On the microbial metabolism of the tartaric acids isomers. Dissertation, University of Delft
Nakas JP, Schaedle M, Parkinson CM, Coonley CE, Tanenbaum SW (1983) System development for linked-fermentation products of solvents from algal biomass. Appl Environ Microbiol 46:1017–1023
Pfennig N, Lippert KD (1966) Über das Vitamin B12-Bedürfnis phototropher Schwefelbakterien. Arch Microbiol 55:245–256
Ruch FE, Lengeler J, Lin ECC (1974) Regulation of glycerol catabolism in Klebsiella aerogenes. J Bacteriol 119:50–56
Schmidt K, Liaanen-Jensen S, Fishkes M (1963) Die Carotinoide der Thiorhodaceae. Arch Microbiol 46:117–126
Schütz H, Radler F (1984) Anaerobic reduction of glycerol to propanediol-1,3 by Lactobacillus brevis and Lactobacillus buchneri. Syst Appl Microbiol 5:169–178
Streekstra H, Teixeira De Mattos MJ, Neijssel OM, Tempest OW (1987) Overflow metabolism during anaerobic growth of Klebsiella aerogenes NCTC 418 on glycerol and dihydroxyacetone in chemostat culture. Arch Microbiol 47:268–275
Toraya T, Ushio K, Fukui S, Hogenkamp HPC (1977) Studies on the mechanism of the adenosylcobalamin-dependent dioldehydratase reaction by the use of analogs of the coenzyme. J Biol Chem 252:963–970
Author information
Authors and Affiliations
Additional information
Correspondence to: G. Gottschalk
Rights and permissions
About this article
Cite this article
Boenigk, R., Bowien, S. & Gottschalk, G. Fermentation of glycerol to 1,3-propanediol in continuous cultures of Citrobacter freundii . Appl Microbiol Biotechnol 38, 453–457 (1993). https://doi.org/10.1007/BF00242936
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00242936