Abstract
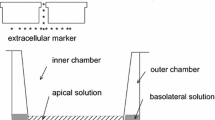
We have previously reported that the isolated frog corneal epithelium (a Cl−-secreting epithelium) has a large diffusional water permeability (Pdw ≈ 1.8×10−4 cm/s). We now report that the presence of Cl− in the apical-side bathing solution increases the diffusional water flux, Jdw (in both directions) by 63% from 11.3 to 18.4 μl min−1 · cm−2 with 60 mm [Cl] exerting the maximum effect. The presence of Cl− in the basolateral-side bathing solution had no effect on the water flux. In Cl−-free solutions amphotericin B increased Jdw by 29% but only by 3% in Cl−-rich apical-side bathing solution, suggesting that in Cl−-rich apical side bathing solution, the apical barrier is no longer rate limiting. Apical Br− (75 mm) also increased Jdw by 68%. The effect of Cl− on Jdw was observed within 1 min after its addition to the apicalside bathing solution. HgCl2 (0.5 mm) reduced the Cl−-increased Pdw by 31%. The osmotic permeability (Pf) was also measured under an osmotic gradient yielding values of 0.34 and 2.88 (x 10−3 cm/s) in Cl−-free and Cl−-rich apical-side bathing solutions respectively. It seems that apical Cl−, or Cl− secretion into the apical bath could activate normally present but inactive water channels. In the absence of Cl−, water permeability of the apical membrane seems to be limited to the permeability of the lipid bilayer.
Similar content being viewed by others
References
Bentley, P.J., Candia, O.A. 1975. Effects of amphotericin B on ionic transport and sodium permeability of the toad lens. Am. J. Physiol. 229:1520–1525
Candia, O.A. 1972. Ouabain and sodium effects on chloride fluxes across the isolated bullfrog cornea. Am. J. Physiol. 223:1053–1057
Candia, O.A. 1973. Short-circuit current related to active transport of chloride in frog cornea: effects of furosemide and ethacrynic acid. Biochim. Biophys. Acta 298:1011–1014
Candia, O.A. 1976. Fluid and chloride transport by the epithelium of the isolated frog cornea. Fed. Proc. 35 (Part 1):703
Candia, O.A. 1984. Secretion of chloride by the frog cornea. In: Chloride Transport Coupling in Biological Membranes and Epithelia. G.A. Gerencser, editor. pp. 393–414. Elsevier, North-Holland
Candia, O.A., Bentley, P.J., Cook, P.I. 1974. Stimulation by amphotericin B of active Na transport across amphibian cornea. Am. J. Physiol. 226:1438–1444
Candia, O.A., Mia, A.J., Yorio, T. 1993. Influence of filter supports on transport characteristics of cultured A6 kidney cells. Am. J. Physiol. 265:C1479-C1488
Candia, O.A., Montoreano, R., Podos, S.M. 1977. Effect of the ionophore A23187 on chloride transport across isolated frog cornea. Am. J. Physiol. 233:F94-E101
Chalfie, M., Neufeld, A.H., Zadunaisky, J.A. 1972. Action of epinephrine and other cyclic AMP-mediated agents on the chloride transport of the frog cornea. Invest. Ophthalmol. 11:644–650
Clarke, L.L., Grubb, B.R., Gabriel, S.E., Smithies, O., Koller, B.H., Boucher, R.C. 1992. Defective epithelial chloride transport in a gene-targeted mouse model of cystic fibrosis. Science 257:1125–1128
Finkelstein. A. 1976. Nature of the water permeability increase induced by antidiuretic hormone (ADH) in toad urinary bladder and related tissues. J. Gen. Physiol. 68:137–143
Fischbarg, J. 1992. The biology of the cornea. In: Fundamentals of medical cell biology. Vol. 6. E.E. Bittar, editor. p. 151. JAI Press
Fischbarg, J., Lim, J.J. 1974. Role of cations, anions and carbonic anhydrase in fluid transport across rabbit corneal endothelium. J. Physiol. 241:647–675
Fischbarg, J., Montoreano, R. 1982. Osmotic permeabilities across corneal endothelium and antidiuretic hormone-stimulated toad urinary bladder structures. Biochim. Biophys. Acta 690:207–214
Hasegawa, H., Skach, W., Baker, O., Calayag, M.C., Lingappa, V., Verkman, A.S. 1992. A multifunctional aqueous channel formed by CFTR. Science 258:1477–1479
Hays, R.M., Leaf, A. 1962. Studies on the movement of water through the isolated toad bladder and its modification by vasopressin. J. Gen. Physiol. 45:905–914
Hodson, S., Miller, F. 1976. The bicarbonate ion pump in the endothelium which regulates the hydration of the rabbit cornea. J. Physiol. 263:271–302
Holz, R., Finkelstein, A. 1970. The water and nonelectrolyte permeability induced in thin lipid membranes by the polyene antibiotics nystatin and amphotericin B. J. Gen. Physiol. 56:125–145
Klyce, S.D. 1975. Transport of Na+, Cl−, and water by the rabbit corneal epithelium at resting potential. Am. J. Physiol. 228:1446–1452
Lichtenstein, N.S., Leaf, A. 1965. Effect of amphotericin B on the permeability of the toad bladder. J. Clin. Invest. 44:1328–1342
Maurice, D.M. 1972. The localization of the fluid pump in the cornea. J. Physiol. 221:43–54
Neilsen, S., Smith, B.L., Christenson, E.I., Knepper, M.A., Agre, P. 1992. CHIP28 water channels are localized in constitutively water permeable segments of the nephron. J. Cell Biol. 120:371–383
Parisi, M., Candia, O.A., Alvarez, L. 1980. Water permeability of the toad corneal epithelium: the effects of pH and amphotericin B. Pfluegers Arch. 383:131–136
Preston, G.M., Carrol, T.P., Guggino, W.B., Agre, P. 1992. Appearance of water channels in Xenopus oocytes expressing red cell CHIP28 protein. Science 256:385–387
Van Hoek, A.N., Verkman, A.S. 1992. Functional reconstitution of the isolated erythrocyte water channel CHIP28. J. Biol. Chem. 267:18267–18269
Verkman, A.S. 1989. Mechanisms and regulation of water permeability in renal epithelia. Am. J. Physiol. 257:C837-C850
Zadunaisky, J.A. 1966. Active transport of chloride in frog cornea. Am. J. Physiol. 211:506–512
Author information
Authors and Affiliations
Additional information
This work was supported by National Eye Institute grants EY-00160 and EY-01867.
Rights and permissions
About this article
Cite this article
Candia, O.A., Zamudio, A.C. Chloride-activated water permeability in the frog corneal epithelium. J. Membarin Biol. 143, 259–266 (1995). https://doi.org/10.1007/BF00233454
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF00233454