Abstract
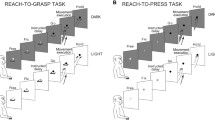
We compared neuronal activity in the dorsal premotor cortex (PMd), ventral premotor cortex (PMv), and prefrontal (PF) cortex of two rhesus monkeys. The behavioral design was a variant of the instructed delay task which established that: (1) a given visual stimulus could, on different trials, instruct different limb movements and (2) several different visual stimuli could instruct the same movement. Neurons in all frontal areas displayed the often replicated activity patterns that occur during instructed delay tasks, including phasic increases after instruction stimuli (signal-related activity), tonic discharge during an instructed delay period (set-related activity), and phasic premovement discharge (movementrelated activity). For signal-, set-, and movement-related activity, the majority of neurons in PMd (51–64%), but only a minority in PF (16–18%) and PMv (32–40%), showed activity levels that significantly depended on the action instructed by that stimulus rather than simply the characteristics of the stimulus per se. Thus, most PMd activity, including the aspects that most resembled a sensory response, reflected factors in addition to the signal. Taken together with the results of related studies, it seems most likely that these other factors are dominated by the motor instructional significance of the stimulus. In addition, many neurons (17–37%) in all examined areas showed activity that significantly depended on which of various stimuli guided the same movement. This finding shows that, in those frontal areas, neuronal activity can be affected by both the action to be taken and the events guiding that action.
Similar content being viewed by others
References
Alexander GE, Crutcher MD (1990) Neural representation of the target (goal) of visually guided arm movements in three motor areas of the monkey. J Neurophysiol 64:164–178
Boch RA, Goldberg ME (1989) Participation of prefrontal neurons in the preparation of visually guided eye movements in the rhesus monkey. J Neurophysiol 61:1064–1084
Boussaoud D, Wise SP (1993) Primate frontal cortex: neuronal activity following attentional vs intentional cues. Exp Brain Res 95:28–40
Boussaoud D, Barth TM, Wise SP (1993) Effects of gaze on apparent visual responses of frontal cortex neurons. Exp Brain Res 93:423–434
Caminiti R, Johnson PB, Urbano A (1991) Making arm movements within different parts of space: the premotor and motor cortical representation of a coordinate system for reaching to visual targets. J Neurosci 11:1182–1197
Crammond DJ, Kalaska JF (1990) Cortical neuronal activity recorded in a delay task that dissociates location of cue stimulus and movement endpoint. Soc Neurosci Abstr 16:423
di Pellegrino G, Wise SP (1991) A neurophysiological comparison of three distinct regions of the primate frontal lobe. Brain 114:951–978
di Pellegrino G, Wise SP (1993) Visuospatial vs visuomotor activity in the premotor and prefrontal cortex of a primate. J Neurosci 13:1227–1243
Funahashi S, Bruce CJ, Goldman-Rakic PS (1989) Mnemonic coding of visual space in the monkey's dorsolateral prefrontal cortex. J Neurophysiol 61:331–349
Funahashi S, Bruce CJ, Goldman-Rakic PS (1990) Visuospatial coding in primate prefrontal neurons revealed by oculomotor paradigms. J Neurophysiol 63:814–831
Funahashi S, Bruce CJ, Goldman-Rakic PS (1991) Neuronal activity related to saccadic eye movements in the monkey's dorsolateral prefrontal cortex. J Neurophysiol 65:1464–1483
Gentilucci M, Fogassi L, Luppino G, Matelli M, Camarda R, Rizzolatti G (1988) Functional organization of inferior area 6 in the macaque monkey. I. Somatotopy and the control of proximal movements. Exp Brain Res 71:475–490
Godschalk M, Lemon RN, Kuypers HGJM, van der Steen J (1985) The involvement of monkey premotor cortex neurones in preparation of visually cued arm movements. Behav Brain Res 18:143–157
Goodale MA, Milner AD, Jakobson LS, Carey DP (1991) A neurological dissociation between perceiving objects and grasping them. Nature 349:154–156
Henry FM, Rogers DE (1960) Increased response latency for complicated movements and a “memory drum” theory of neuromotor reaction. Res Q 31:448–458
Kubota K, Hamada I (1978) Visual tracking and neuron activity in the postarcuate area in monkeys. J Physiol (Paris) 74:297–312
Kurata K, Tanji J (1986) Premotor cortex neurons in macaques: activity before distal and proximal forelimb movements. J Neurosci 6:403–411
Kurata K, Wise SP (1988a) Premotor cortex of rhesus monkeys: set-related activity during two conditional motor tasks. Exp Brain Res 69:327–343
Kurata K, Wise SP (1988b) Premotor and supplementary motor cortex in rhesus monkeys: neuronal activity during externally- and internally-instructed motor tasks Exp Brain Res 72:237–248
Lecas J-C, Requin J, Anger C, Vitton N (1986) Changes in neuronal activity of the monkey precentral cortex during preparation for movement. J Neurophysiol 56:1680–1702
Lurito JT, Georgakopoulos T, Georgopoulos AP (1991) Cognitive spatial motor processes. 7. The making of movements at an angle from a stimulus direction: studies of motor cortical activity at the single cell and population levels. Exp Brain Res 87:562–580
Meeres SL, Graves RE (1990) Localization of unseen visual stimuli by humans with normal vision. Neuropsychologia 28:1231–1237
Mitz AR, Godschalk M, Wise SP (1991) Learning-dependent neuronal activity in the premotor cortex of rhesus monkeys. J Neurosci 11:1855–1872
Moran J, Desimone R (1985) Selective attention gates visual processing in the extrastriate cortex. Science 229:782–784
Mushiake H, Inase M, Tanji J (1991) Neuronal activity in the primate premotor, supplementary, and precentral motor cortex during visually guided and internally determined sequential movements. J Neurophysiol 66:705–718
Niki H, Watanabe M (1976) Prefrontal unit activity and delayed response: relation of cue location versus direction of response. Brain Res 105:79–88
Passingham RE (1988) Premotor cortex and preparation for movement. Exp Brain Res 70:590–596
Riehle A, Requin J (1989) Monkey primary motor and premotor cortex: single-cell activity related to prior information about direction and extent of an intended movement. J Neurophysiol 61:534–549
Rizzolatti G, Scandolara C, Matelli M, Gentilucci M (1981) Afferent properties of periarcuate neurons in macaque monkeys. 2. Visual responses. Behav Brain Res 2:147–163
Rizzolatti G, Camarda R, Fogassi L, Gentilucci M, Luppino G, Matelli M (1988) Functional organization of inferior area 6 in the macaque monkey. II. Area F5 and the control of distal movements. Exp Brain Res 71:491–507
Spitzer H, Richmond BJ (1991) Task difficulty: ignoring, attending to, and discriminating a visual stimulus yield progressively more activity in inferior temporal neurons. Exp Brain Res 83:340–348
Tanji J, Kurata K (1982) Comparison of movement-related activity in two cortical motor areas of primates. J Neurophysiol 48:633–653
Tanji J, Okano K, Sato KC (1988) Neuronal activity in cortical motor areas related to ipsilateral, contralateral, and bilateral digit movements of the monkey. J Neurophysiol 60:325–343
Taylor JL, McCloskey DI (1990) Triggering of preprogrammed movements as reactions to masked stimuli. J Neurophysiol 63:439–446
Vaadia E, Benson DA, Hienz RD, Goldstein MH (1986) Unit study of monkey frontal cortex: active localization of auditory and of visual stimuli. J Neurophysiol 56:934–952
Vaadia E, Kurata K, Wise SP (1988) Neuronal activity preceding directional and nondirectional cues in the premotor cortex of rhesus monkeys. Somatosensory Motor Res 6:207–230
Watanabe M (1986a) Prefrontal unit activity during delayed conditional go/no-go discrimination in the monkey. 1. Relation to the stimulus. Brain Res 382:1–14
Watanabe M (1986b) Prefrontal unit activity during delayed conditional go/no-go discrimination in the monkey. II. Relation to go and no-go responses. Brain Res 382:15–27
Watanabe M (1990) Prefrontal unit activity during associative learning in the monkey. Exp Brain Res 80:296–309
Watanabe M (1992) Frontal units of the monkey coding the associative significance of visual and auditory stimuli. Exp Brain Res 89:233–247
Weinrich M, Wise SP (1982) The premotor cortex of the monkey. J Neurosci 2:1329–1345
Weinrich M, Wise SP, Mauritz K-H (1984) A neurophysiological analysis of the premotor cortex of the monkey. Brain 107:385–414
Weiskrantz L, (1989) Blindsight: a case study and implications. Oxford University Press, Oxford
Wise SP (1985) The primate premotor cortex fifty years after Fulton. Behav Brain Res 18:79–88
Wise SP, Boussaoud D (1992) Primate frontal cortex: effect of instructional significance on activity following identical visual cues. Soc Neurosci Abstr 18:501
Wise SP, Weinrich M, Mauritz K-H (1983) Motor aspects of cue-related neuronal activity in premotor cortex of the rhesus monkey. Brain Res 260:301–305
Wise SP, di Pellegrino G, Boussaoud D (1992) Primate premotor cortex: dissociation of visuomotor from sensory signals. J Neurophysiol 68:969–972
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Boussaoud, D., Wise, S.P. Primate frontal cortex: effects of stimulus and movement. Exp Brain Res 95, 28–40 (1993). https://doi.org/10.1007/BF00229651
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00229651