Abstract
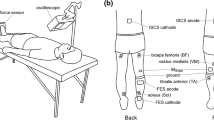
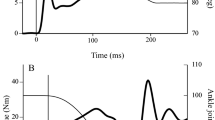
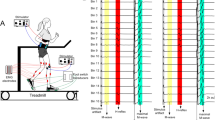
The size of soleus H-reflexes and short-latency stretch reflexes was measured at different levels of plantar flexion or co-contraction (simultaneous activation of dorsi- and plantar flexors) in seven healthy subjects. In four of seven subjects the short-latency stretc reflex was smaller during weak co-contraction than during isolated plantar flexion at matched background electromyogram (EMG) levels in the soleus muscle. In three of these four subjects the stretch reflex was larger during strong co-contraction than during plantar flexion, whereas it had the same size during the two tasks in the last subject. In the remaining subjects the stretch reflex either had the same size or was larger at all levels of co-contraction than at similar levels of plantar flexion. In contrast, the H-reflex was found to decrease with co-contraction at all contraction levels in all subjects. The decrease in the reflexes during weak co-contraction might be caused by presynaptic inhibition of Ia afferents. It is unclear why only the H-reflex decreased during strong co-contraction. The stiffness of the ankle joint was measured from the torque increment following the stretch of the plantar flexors divided by the stretch amplitude. In all subjects the total stiffness of the ankle joint was larger during strong co-contraction than during plantar flexion of similar strength. The stiffness was smaller during weak co-contraction than during weak plantar flexion in three out of seven subjects. The medial gastrocnemius muscle was more active at a given level of soleus activity during the co-contraction task than during the isolated plantar flexion task. It is suggested that the increase in the stiffness during co-contraction as compared to isolated plantar flexion was mainly due to the mechanical contribution of the activity in the tibialis anterior and medial gastrocnemius muscles. The decrease in stiffness during weak co-contraction was, in contrast, most likely mainly caused by modulation of reflex stiffness.
Similar content being viewed by others
References
Akazawa K, Milner TE, Stein RB (1983) Modulation of reflex EMG and stiffness in response to stretch of human finger muscle. J Neurophysiol 49:16–27
Burke D, Gandevia SC, McKeon B (1984) Monosynaptic and oligosynaptic contributions to human ankle jerk and H-reflex. J Neurophysiol 52:435–448
Carter RR, Crago PE, Gorman PH (1993) Nonlinear stretch reflex interaction during cocontraction. J Neurophysiol 69:943–952
Crago PE, Houk JC, Hasan Z (1976) Regulatory actions of human stretch reflex. J Neurophysiol 39:925–935
Crone C, Hultborn H, Mazieres L, Morin C, Nielsen J, Pierrot-Deseilligny E (1990) Sensitivity of monosynaptic test reflexes to facilitation and inhibition as a function of the test reflex size: a study in man and the cat. Exp Brain Res 81:35–445
Datta AK, Farmer SF, Stephens JA (1991) Central nervous pathways underlying synchronization of human motor unit firing studied during voluntary contractions. J Physiol (Lond) 432:401–425
Feldman AG (1980) Superposition of motor programs. I. Rythmic forearm movements in man. Neuroscience 5:81–90
Hoffer JA, Andreassen S (1981) Regulation of soleus muscle stiffness in premammilary cats: intrinsic stiffness and reflex components. J Neurophysiol 45:267–285
Hulliger M, Dürmüller N, Prochazka A, Trend P (1989) Flexible fusimotor control of muscle spindle feedback during a variety of natural movements. Prog Brain Res 80:87–103
Humphrey DR, Reed DJ (1983) Separate cortical systems for control of joint movement and joint stiffness: reciprocal activation and coactivation of antagonist muscles. In: Desmedt JE (ed) Motor control mechanisms in health and disease. Raven New York, pp 347–372
Llewellyn M, Yang JF, Prochazka A (1990) Human H-reflexes are smaller in difficult beam walking than in normal treadmill walking. Exp Brain Res 83:22–28
Nichols TR, Houk JC (1976) Improvements in linearity and regulation of stiffness that results from actions of stretch reflex. J Neurophysiol 39:119–142
Nichols TR (1989) The organization of heterogenic reflexes among muscles crossing the ankle joint in the decerebrate cat. J Physiol (Lond) 410:463–477
Nielsen J, Kagamihara Y (1992) Regulation of reciprocal Ia inhibition during co-contraction of antagonistic muscles in man. J Physiol (Lond) 456:373–391
Nielsen J, Kagamihara Y (1993) Regulation of presynaptic inhibition during co-contraction of antagonitsic muscles in man. J Physiol (Lond) 464:575–593
Pierrot-Deseilligny E, Morin C, Bergego C, Tankov N (1981) Pattern of group I fibre projections from ankle flexor and extensor muscles in man. Exp Brain Res 42:337–350
Schieppati M, Romano C, Gritti I (1990) Convergence of Ia fibres from synergistic and antagonistic muscles onto interneurones inhibitory to soleus in humans. J Physiol (Lond) 431:365–377
Sinkjær T, Hoffer JA (1990) Factors determining segmental reflex action in normal and decerebrate cats. J Neurophysiol 64:1652–1635
Smith AM (1981) The co-activation of antagonist muscles. Can J Physiol Pharmacol 59:733–747
Sinkjær T, Toft E, Andreassen S, Hornemann BC (1988) Muscle stiffness in human ankle dorsiflexors: areflexive and reflex components. J Neurophysiol 60:1110–1121
Toft E, Sinkjær T, Andreassen S, Larsen K (1991) Mechanical and electromyographic responses to stretch of the human ankle extensors. J Neurophysiol 65:1402–1410
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Nielsen, J., Sinkjær, T., Toft, E. et al. Segmental reflexes and ankle joint stiffness during co-contraction of antagonistic ankle muscles in man. Exp Brain Res 102, 350–358 (1994). https://doi.org/10.1007/BF00227521
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00227521