Summary
The development of rheological models to predict creep has led to the derivation of quite complex equations that can predict creep reasonably accurately. However, these models are conceptual and are not based on a fundamental understanding of the actual deformation processes occurring within the material. The concept of modelling creep using a chemical kinetic approach is one that attempts to understand creep in wood at a molecular level and, from this, to develop models that more accurately predict creep deflections.
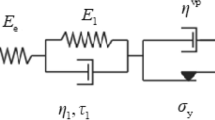
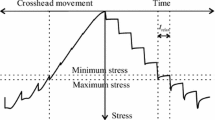
This paper presents two models developed from chemical kinetic theory, that describe the time-dependant deformation of wood. The validity of applying these models to experimental data has been assessed by stress relaxation tests on thin samples of Sequoia sempervirens.
Two stages of experimentation were carried out. In stage 1, both models were applied to the results of stress relaxation tests on 6 samples. Similar values of activation energy and activation volume were calculated by both models and a single energy barrier was found to dominate the deformation process.
In stage 2, the effect of varying the initial applied stress on activation energy and activation volume was assessed by carrying out stress relaxation tests at stress levels of 25%, 30% and 35% of the short-term strength. Values of activation energy and activation were found to increase as the applied stress level decreased.
Both models describe the time-dependent behaviour of wood well, however their ability to predict long-term creep deflections may be limited. Future work will develop these models further in order to improve long-term creep prediction and then apply them to the results of both creep and stress relaxation tests at a variety of stress levels and moisture contents in order to test their validity.
Similar content being viewed by others
Abbreviations
- A:
-
reaction rate constant times the concentration of flow units
- E1 :
-
elastic modulus of elastic component
- E2 :
-
elastic modulus of viscous component
- ΔG:
-
Gibb's free energy
- h:
-
Planck's constant = 4.135×10-15 eV sec-1
- k:
-
Boltzmann's constant = 8.616×10-5 eVK
- k′:
-
rate constant
- R:
-
gas constant = 1.987 calK-1 mol-1
- t:
-
time
- T:
-
absolute temperature
- Vh :
-
activation volume
- Vm :
-
volume of cell wall element
- ɛ:
-
strain
- ϱ:
-
concentration of flow units
- σ:
-
mean stress
- τ:
-
stress on all viscous elements
References
Caulfield, D. F. (1985) A chemical kinetics approach to duration of load problem in wood. Wood and Fibre Science 17(4): 504–521
Flowers, B. H.; Mendoza, E. (1970) Properties of matter. Chichester. John Wiley and Sons Ltd.
Glasstone, S., Laidler, K. J., Eyring, H. (1941) The theory of rate processes. McGraw-Hill Book Co. Inc., New York
Grossman, P. U. A.; Kingston, R. S. T. (1954) Creep and stress relaxation of wood during bending. Australian Journal of Applied Science 5(4): 403–417
Kingston, R. S.; Clarke, L. N. (1961) Some aspects of the rheological behaviour of wood. ii. Analysis of creep data by reaction rate and thermodynamic methods. Australian Journal of Applied Science 12(2): 227–240
Moore, W. J. (1972) Physical chemistry. 5th edition, Longman, London
Pierce, C. B.; Dinwoodie, J. M. (1977) Creep in chipboard. Part 1: Fitting 3- and 4-element response curves to creep data. J. Materials Science 12: 1955–1960
Pierce, C. B.; Dinwoodie, J. M. (1985) Creep in chipboard. Part 5: An Improved model for prediction of creep deflection. Wood Sci. Technol. 19: 83–91
Van der Put, T. A. C. M. (1989) Deformation and damage processes in wood. Ph.D. thesis, Delft University Press
Van der Weil, A. (1984) Time dependant deformation of wood description of stress relaxation of wood in bending by the method of deformation kinetics. Rapport 4-84-4. Onderzoek HA-17, Stevin-Laboratorium van de Afdeling der Civiele Technick der Technische Hgogeschool
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Bonfield, P.W., Mundy, J., Robson, D.J. et al. The modelling of time-dependant deformation in wood using chemical kinetics. Wood Sci.Technol. 30, 105–115 (1996). https://doi.org/10.1007/BF00224962
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00224962