Abstract
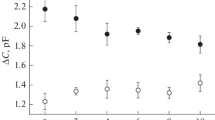
Tripositive-pyrophosphate [M(III)-PPi] complexes were used to investigate the role of free divalent cations on the membrane-bound pyrophosphatase. Divalent cations remain free and the M(III)-PPi complexes were employed as substrates. Formation of a La-PPi complex was studied by fluorescence, and the fact that Zn2+ and Mg2+ remain free in the solution was validated. Hydrolysis of La-PPi is stimulated by the presence of fixed concentrations of free Mg2+ or Zn2+ and this stimulation depends on the concentration of the cations when the La-PPi complex is fixed. The divalent cation stimulation order is Zn2+ > Co2+ > Mg2+ > Mn2+ > Ca2+ (at 0.5 mm of free cation). With different M(III)-PPi complexes, Zn2+ produces the same K m, for all the complexes and Mg2+ stimulates with a different K m. The results suggest that both Mg2+ and Zn2+ activate the membrane-bound pyrophosphatase but through different mechanisms.
Similar content being viewed by others
References
Ames BN. 1966 Assay of inorganic phosphate, total phosphate and phosphatases. Methods Enzymol VIII, 115–118.
Baltscheffsky M. 1978 Photosynthetic phosphorylation. In: Clayton RK, Sistrom, WR, eds. The Photosynthetic Bacteria. New York: Plenum Press; 595–613.
Baltscheffsky H, von Stedingk LV, Heldt HW, Klingenberg M. 1966 Inorganic pyrophosphate: formation in bacterial photophosphorylation. Science 153, 1120–1124.
Celis H, Romero I. 1987 The phosphate pyrophosphate exchange and hydrolytic reactions of membrane-bound pyrophosphatase of Rhodospirillum rubrum: effects of pH and divalent cations. J Bioenerg Biomembr 19, 255–272.
Celis H, Romero I, Gómez-Puyou A. 1985 The phosphate-pyrophosphate exchange and hydrolytic reaction of the membrane-bound pyrophosphatase of Rhodospirillum rubrum: effects of Mg2+, phosphate and pyrophosphate. Arch Biochem Biophys 236, 766–774.
Cohen-Bazire G, Sistrom WR, Stainer RY. 1957 The kinetic studies of pigment synthesis by non-sulfur purple bacteria. J Cell Comp Physiol 49, 25–68.
Heinonen JK, Honkasalo SH, Kukko EI. 1981 A method for the concentration and for the colorimetric determination of nanomoles of inorganic pyrophosphate. Anal Biochem 117, 293–300.
Lahti R. 1983 Microbial inorganic pyrophosphatases. Microbiol Rev 47, 169–179.
Lowry OH, Rosebrough NJ, Farr AL, Randal RJ. 1951 Protein measurements with the folin phenol reagent. J Biol Chem 193, 265–275.
Martell A, Sillén LG. 1971 Stability Constants of Metal-Ion Complexes, Suppl. 1. Special Publication 25. London: The Chemical Society.
Moeller T. 1972 Complexes of the lanthanides. In: Bagnall KW, ed. Lanthanides and Actinides. MTP International Review of Science. London: Butterworths; 275–298.
Randahl H. 1979 Characterization of the membranebound inorganic pyrophosphatase in Rs. rubrum. Eur J Biochem 102, 251–256.
Smith RM, Martell AE. 1976 Critical Stability Constants. Vol 4: Inorganic Complexes. New York: Plenum Press.
Sosa A, Ordaz H, Romero I, Celis H. 1992 Mg2+ is an essential activator of hydrolytic activity of membranebound pyrophosphatase of Rhodosopirillum rubrum. Biochem J 283, 561–566.
Sumner JB. 1944 A method for the colorimetric determination of phosphorous. Science 100, 413–415.
Ting S, Dunaway-Marino D. 1984 Investigation of the role of the substrate metal ion in the yeast inorganic pyrophosphatase reaction. FEBS Lett 165, 251–253.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Velázquez, I., Celis, H. & Romero, I. Regulation of divalent cations of the membrane-bound pyrophosphatase of Rhodospirillum rubrum, as shown by the hydrolysis of tripositive-pyrophosphate complexes. Biometals 6, 143–148 (1993). https://doi.org/10.1007/BF00205852
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00205852