Abstract
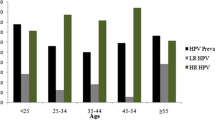
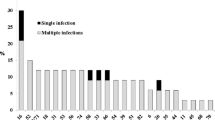
Seventy-seven women with normal cervical cytology on routine visit to a family planning clinic in Nairobi, Kenya, were analysed for genital human papillomavirus (HPV) types by polymerase chain reaction (PCR). We applied a general primer pair (GP60/GP124) recognising sequences conserved among HPV types 6, 11, 16, 18, 31 and 33. Of the 77 specimens tested 15 (19.5%) proved to be positive for genital HPV. Amplification products were examined for the presence of high-risk HPV types by Slot-blot hybridization. Out of the 15 PCR-positive samples, 4 were positive for HPV 16,3 for HPV 18, while 1 contained both HPV 16 and 33. HPV DNA prevalence in this group of women from a “high-risk” area is similar to that in “low-risk” Swedish women but much lower than in cervical cancer samples from the same region.
Similar content being viewed by others
References
Chan WK, Klock G, Bernard HU (1989) Progesterone and glucocorticoid response elements occur in the long control region of several human papillomaviruses involved in oncogenital neoplasia. J Virol 63:4417–4421
Czeglédy J, Gergely L, Endrödi J (1989) Detection of human papillomavirus deoxyribonucleic acid by filter in situ hybridization during pregnancy. J Med Virol 28:250–254
Devereux J, Haeberli P, Smithies O (1984) A comprehensive set of sequence analysis program for the VAX. Nucleic Acids Res 12:385–387
Evander M, Wadell G (1991) A general primer pair amplification and detection of genital human papillomavirus types. J Virol Methods 31:239–250
Higuchi R (1989) Rapid efficient DNA extraction for PCR from cells or blood. Amplifications. A forum for PCR users. Perkin Elmer Cetus 2:1–3
Pao CC, Lin CY, Maa JS, Lai CH, Wu SY, Soong YK (1990) Detection of human papillolmaviruses in cervicovaginal cells using polymerase chain reaction. J Infect Dis 161:113–115
Rogo KO (1989) Mortality in acute gynecology; a developing country perspective. Int J Gynecol Obstet 30:343–347
Rogo KO, Evander M, Bjersing L, Stendahl U, Wadell G (1991) Double HPV infection in African cervical cancer detected by polymerase chain reaction. Anticancer Res 11:169–174
Saiki RK, Scharf S, Faloona F, Mullis KB, Horn GT, Erlich HA, Arnheim N (1985) Enzimatic amplification of β-globin genomic sequences and restriction site analysis for diagnosis of sickle cell anemia. Science 230:1350–1354
Schwarz E, Freese UK, Gissmann L, Mayer W, Roggenbuck B, Stremlau A, zur Hausen H (1985) Structure and transcription of human papillomavirus sequences in cervical carcinoma cells Nature 314:111–114
Tanaka A, Noda J, Yajuma H, Hatanaka M, Ho Y (1989) Identification of transforming gene of human papillomavirus type 16. J Virol 63:1465–1469
Van der Brule AJC, Claas ECJ, du Maine M, Melchers WJG, Helmerhorst T, Quint WGV; Lindeman J, Meijer CJLM, Walboomers JMM (1989) Use of anticontamination primers in the polymerase chain reaction for the detection of human papillomavirus genotypes in cervical scrapes and biopsies. J Med Virol 29:20–27
Walker J, Bloss JD, Liao SY, Berman M, Bergen S, Wilczynski SP (1989) Human papillomavirus genotype as a prognostic indiqator in carcinoma of the uterine cervix. Obstet Gynecol 74:781–785
Xiao X, Cao M, Miller TR, Yen TSB (1988) Papillomavirus DNA in cervical carcinoma specimens from Cetral China. Lancet i:902
zur Hausen H, Schneider A (1987) The role of papillomaviruses in human anogenital cancer. In: Salzman NP, Howley PM (eds) The papovaviridae, vol 2. The papillomaviruses. Plenum Press, New York, pp 245–263
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Czeglédy, J., Rogo, K.O., Evander, M. et al. High-risk human papillomavirus types in cytologically normal cervical scrapes from Kenya. Med Microbiol Immunol 180, 321–326 (1992). https://doi.org/10.1007/BF00191552
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00191552