Summary
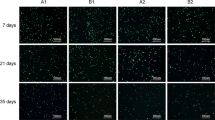
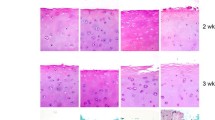
We have investigated the viability and function of cells in cartilage slices after various methods of preservation, and have examined the viability of cells by measuring the incorporation of Na2 35SO4 at different concentrations, temperatures and times of exposure to cryopreservatives. We have assessed the viability of cells when exposed to prefreezing temperatures, and after preservation at −80° C. The best survival rate was found to be with a concentration of cryopreservatives of approximately 10%, with pre-freeze exposure for four hours at 4° C. In the stage cooling technique, the best initial cooling was at −30° C for 30 minutes, followed by rapid cooling of the cartilage to −80° C. The best survival rate for cryopreserved cartilage in 10% DMSO was, on average, 19% in intact slices and 34% when holes had been made in the slices. Proteoglycan synthesis after thawing appeared normal, and proteoglycan labelled after 48 hours in culture also showed a normal pattern.
Résumé
Nous avons étudié la viabilité et la fonction cellulaire de coupes de cartilage après diverses méthodes de conservation et nous avons évalué la viabilité des cellules en mesurant l'absorption du Na2 35SO4 à différentes concentrations, températures et durées d'exposition aux cryo-préservateurs. Nous avons évalué la viabilité des cellules exposées aux températures de pré-congélation et après conservation à −80° C. Nous avons trouvé que le meilleur pourcentage de survie était obtenu avec une concentration de cryo-préservateurs d'environ 10%, avec une pré-congélation de 4 heures à 4° C. Dans la technique de refroidissement par étapes, le meilleur refroidissement initial est de −30° C pendant 30 minutes, suivi d'un refroidissement rapide du cartilage jusqu'a −80° C. Le meilleur taux de survie pour le cartilage cryo-préservé dans 10% de DMSO a été, en moyenne, de 19% dans les coupes intactes et de 34% quand des perforations ont été pratiquées dans les coupes. La synthèse du protéoglycan après décongélation paraît normale et le protéoglycan marqué, après une culture de 48 heures, s'avère également normal.
Similar content being viewed by others
References
Bourongcle BA (1967) Preservation of normal and leukemic cells with dimethyl sulfoxide at -80° C. Cryobiology 3: 445–455
Brighton CT, Shadle CA, Jimenez SA, Irwin JT, Lane JM, Lipton M (1979) Articular cartilage preservation and storage. 1. Application of tissue culture techniques to the storage of viable articular cartilage. Arthrit Rheumatol 22: 1093–1101
Curran RC, Gibson T (1956) The uptake of labelled sulphate by human cartilage cells and its use as a test for viability. Proc R Soc London B 144: 572–576
Gibson T (1957) Viability of cartilage after freezing. Proc R Soc London B 147: 528–529
Holtzer H, Abbott J, Lash J, Holtzer S (1960) The loss of phenotypic traits by differenciated cells in vitro. 1. Differenciation of cartilage cells. Proc Nat Acad Sci 46: 1533–1542
Luyet B, Keane J Jr (1955) A critical temperature range apparently characterized by sensitivity of bull semen to high freezing velocity. Biodynamica 7: 281–292
Mankin HJ, Doppelt SH, Sullivan TR, Tomford WW (1982) Osteoarticular and intercalary allograft transplantation in the management of malignant tumors of bone. Cancer 50: 613–630
Meryman HT, Williams RJ, Donglas MSTJ (1977) Freezing injury from “Solution Effects” and its preservation by natural or artificial cryopreservation. Cryobiology 14: 287–302
Pegg DE, Trotman RE (1959) The preservation of human bone marrow at -79° C. A temperaturecontrolled method of two stage cooling. J Clin Pathol 12: 477–482
Prudden TM (1881) Experimental studies on the transplantation of cartilage. Am J Med Sci 82: 360
Sherman JK (1965) Pretreatment with protective substances as a factor in freeze-thaw survival. Cryobiology 1: 298–300
Smith AU (1965) Survival of frozen chondrocytes isolated from cartilage of adult mammals. Nature 205: 782–784
Stulberg CS, Soule HD, Berman L (1958) Preservation of human-epitherial-like and fibroblast-like cell strains at low temperatures. Proc Soc Exper Biol Med 98: 428–431
Tomford W, Fredericks GR, Mankin HJ (1984) Studies on cryopreservation of articular cartilage chondrocytes. J Bone Joint Surg [Am] 66: 253–259
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Kawabe, N., Yoshinao, M. Cryopreservation of cartilage. International Orthopaedics 14, 231–235 (1990). https://doi.org/10.1007/BF00178751
Issue Date:
DOI: https://doi.org/10.1007/BF00178751