Summary
An increase in the opportunistic fungal infections necessitates a design of new more effective and safer antifungal agnets. Triazole alcohols are effective antifungals, but have a risk of teratogenicity associated with them. Therefore, successful design of drugs from this class depends on understanding the structure-activity and structure-teratogenicity relationships in conjunction.
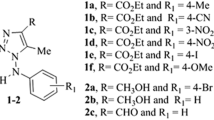
To this end, we applied the Multiple Computer-Automated Structure Evaluation (Multi-CASE) methodology to a study of the relationships between the structures of 71 triazole alcohols and their in vitro antifungal activity, teratogenicity, and therapeutic index. For each end point, several relevant structural descriptors were identified.
A comparative analysis of the Multi-CASE results indicates that cyano, methoxy groups, and ortho-difluorination on the aromatic ring decrease antifungal activity, but not the therapeutic index because of the concomitant negative contribution to teratogenicity. Metabolically deactivating para-substitution in the benzene ring is beneficial for the therapeutic index in agreement with the idea of metabolically induced teratogenicity. Fluorinated para-alkyl substituents are most preferable. The pattern of ortho-substitution in the benzene ring affects both antifungal and teratogenic activity. This suggests that the relative orientation of the benzene ring with respect to the rest of the molecule may play a modulating role.
The Multi-CASE model could correctly predict, a priori, the teratogenicity and antifungal potency of SCH 39304 and ICI 156,066 and be used to optimize the structure and therapeutic index of the latter.
Similar content being viewed by others
References
Graybill, J.R., In Ryley, J.F. (Ed.) Chemotherapy of Fungal Diseases, Springer-Verlag, Berlin, Heidelberg, New York, 1990, pp. 455–475.
Richardson, K., Andrews, R.J., Marriott, M.S. and Troke, P.F., Rec. Adv. Chemother., 3 (1985) 1940.
Humphrey, M.J., Jevons, S. and Tarbit, M.H., Antimicrob. Agents Chemother., 28 (1985) 648.
Hobbs, M.M., Wright, K.A., Perfect, J.R., Tso, C.Y. and Durack, D.T., In 28th Interscience Conference on Antimicrobial Agents and Chemotherapy, Los Angeles, CA, 1988, Abstr. 166.
Grassberger, M.A., Meingassner, J.G. and Schaub, F., Rev. Iber. Micolog., 5 (Suppl. 1) (1988) Abstr. P-153.
Hector, R.F., Rev: Iber. Micolog., 5 (Suppl. 1) (1988) 11 (Abstr. 0–4).
Ryley, J.F., McGregor, S. and Wilson, R.G., Ann. NY Acad. Sci., 544 (1988) 310.
Flint, O.P. and Boyle, E.T., Concepts Toxicol., 3 (1985) 29.
Bechter, R., Hamada, M. and Hakashima, T., Rev. Iber. Micolog., 5 (Suppl. 1) (1988) Abstr. P-149.
Ryley, J.F. and Wilson, R.G., In 22nd Interscience Conference on Antimicrobial Agents and Chemotherapy, Miami Beach, 1982, Abstr. 477.
Flint, O.P. and Boyle, T.F., In Ryley, J.F. (Ed.) Chemotherapy of Fungal Diseases, Springer-Verlag, Berlin, Heidelberg, New York, 1990, pp. 231–248.
Klopman, G., J. Am. Chem. Soc., 106 (1984) 7315.
Klopman, G. and Kalos, A.N., J. Comput. Chem., 6 (5) (1985) 492.
Klopman, G., Quant. Struct.-Act. Relat., 11 (1992) 176.
Klopman, G. and McGonigal, M., J. Chem. Inf. Comput. Sci., 21 (1981) 48.
Nelson, S.D., J. Med. Chem., 25 (1982) 753.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Klopman, G., Ptchelintsev, D. Antifungal triazole alcohols: A comparative analysis of structure-activity, structure-teratogenicity and structure-therapeutic index relationships using the Multiple Computer-Automated Structure Evaluation (Multi-CASE) methodology. J Computer-Aided Mol Des 7, 349–362 (1993). https://doi.org/10.1007/BF00125508
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00125508