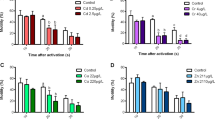
Acetylcholinesterase (AChE) is used as a specific biomarker of the effects of organophosphorous (OP) and carbamate (C) insecticides on the coastal marine environment. Studies of mixtures (by pairs) of five of these substances showed cumulative, synergistic inhibitory effects in all cases. The strongest synergy was observed in organophosphate-carbamate mixtures (OP-C) and the least in mixtures of substances of the same type (OP-OP, C-C). The intensity of the synergistic effect was directly related to the length of time the enzyme was incubated with the inhibitory mixtures. Among the major organic contaminants of the marine environment, DDT and lindane (organochlorines), as well as atrazine and isoproturon, are not AChE inhibitors and had no effect on the inhibitory action of the OP and C insecticides tested. Among contaminants of metallic origin, zinc chloride, cadmium chloride, tributyltin chloride and methylmercury did not inhibit AChE at the concentrations measured in the different marine compartments (water, sediment, living matter). Mercuric chloride and arsenite had a weak inhibitory action in certain organisms. Zinc chloride, cadmium chloride and arsenic enhanced the inhibitory effects of some OP and C insecticides. The dragonet (Callionymus lyra) proved to be a particularly sensitive target species for monitoring pollutant effects.
Similar content being viewed by others
References
Barcelo, D., Sole, M., Durand, G. and Albaiges, J. (1991) Analysis and behaviour of organophosphorous pesticides in a rice crop field. Fresenius J. Anal. Chem. 339, 676–83.
Bocquené, G. and Galgani, F. (1991) Acetylcholinesterase activity in the common prawn (Palaemon serratus) contaminated by carbaryl and phosalone. Ecotoxicol. Environ. Safety 22, 337–45.
Bocquené, G., Galgani, F. and Truquet, P. (1990) Characterisation and assay conditions for the use of AChE activity from several marine species in pollution monitoring. Mar. Environ. Res. 30, 75–89.
Bon, S., Toutant, J.P., Khaled, M. and Massoulié, J. (1988) Amphiphilic and nonamphiphilic forms of Torpedo: II. Electrophoretic variants and phosphatidylinositol phospholipase C-sensitive and insensitive forms. J. Neurochem. 51(3), 776–85.
Burridge, L.E. and Haya, K. (1988) The use of a fugacity model to assess the risk of pesticides on the aquatic environment on Prince Edward Island. Adv. Environ. Sci. Technol. 22, 193–203.
Chang, S. and Opperman, C.H. (1992) Separation and characterization of Heterodera glycines acetylcholinesterase molecular forms. J. Nematol. 22(4), 481–8.
Coppage, D.L. and Matthews, E. (1974) Short-term effects of organophosphate insecticides on cholinesterases of estuarine fishes and pink shrimp. Bull. Environ. Contam. Toxicol. 11(5), 438–88.
Cossa, D., Auger, D., Averty, B., Luçon, M., Masselin, P., Noel, J. and Sanjuan, J. (1989) Atlas des niveaux de concentrations en métaux, métalloïdes et composés organochlorés dans les produits de la pêche côtière française., IFREMER. DERO. 89-10-MR 2–38.
Day, K.E. and Scott, I.M. (1990) Use of acetylcholinesterase activity to detect sublethal toxicity in stream invertebrates exposed to low concentrations of organophosphate insecticides. Aquat. Toxicol. 18, 101–14.
Ellman, G.L., Courtney, K.O., Andrres, V. and Featherstone, R.M. (1961) A new and rapid colorimetric determination of acetylcholinesterase activity. Biochem. Pharmacol. 7, 88–95.
Eto, M. (1974) Organophosphorus pesticides: Organic and Biological Chemistry. CRC Press, Cleveland, OH.
Fournier, D., Cuany, A., Bride, J.M. and Bergé, J.B. (1987) Molecular polymorphism of head acetylcholinesterase from adult houseflies (Musca domestica L.). J. Neurochem. 49 (5), 1455–61.
Gaddum, J.H. (1948) Pharmacology, 3rd edn. Oxford University Press, London.
Galgani, F., Bocquené, G. and Cadiou, Y. (1992a) Evidence of variation in cholinesterase activity in fish along a pollution gradient in the North Sea. Mar. Ecol. Prog. Ser. 91 (1–3), 77–82.
Galgani, F. Cadiou, Y. and Gilbert, F. (1992b) Simultaneous and iterative weighted regression analysis of toxicity tests using a microplate reader. Ecotoxicol. environ Safety 23, 237–43.
Habig, C., DiGiulio, R.T., Nomeir, A.A. and Abou-Donia, M.B. (1986) Comparative toxicity, cholinergic effects and tissue levels of S,S,S-Tri-n-butyl phosphorotrithioate (DEF) to Channel catfish (Ictalurus punctatus) and Blue crab (Callinectes sapidus). Aquat. Toxicol. 9, 193–206.
Habig, C., DiGiulio, R.T. and Abou-Donia, M.B. (1988) Comparative properties of Channel catfish (Ictalurus punctatus) and Blue crab (Callinectes sapidus) acetylcholinesterases. Comp. Biochem. Physiol. 91 C (2), 293–300.
Klaverkampf, J.F. and Hobden, B.R. (1980) Brain acetylcholinesterase inhibition and hepatic activation of acephate and fenitrothion in rainbow trout (Salmo gardeneri). Can. J. Fish. Aquat. Sci. 37, 1450–3.
Lenoir-Rousseaux, J.J., Arpagaus, M. and Toutant, J.P. (1988) Separation and characterization of hydrophilic and detergent-interacting components of brain acetylcholinesterase from Tenebrio molitor L., Comp. Biochem. Physiol. 90 B (1), 29–35.
Michel, P. (1993) L'arsenic en milieu marin. Biogéochimie et écotoxicologie. IFREMER. Repère Océan 4.
Michel, P., Averty, B., Noel, J. and Sanjuan, J. (1992) Evaluation of dissolved and particulate arsenic flux in the Dover Strait., Oceanol. Acta. 16 (5–6), 585–91.
Mulder, J.G. and Bakker, J. (1992) Kinetics of inhibition of two forms of acetylcholinesterase from Panagrellus redivivus by organophosphorous and carbamate compounds. Fund.. Appl. Nematol. 15 (1), 19–23.
Nemcsok, J., Orban, L., Dobber, L. and Szepealussy, J. (1985) Acetylcholinesterase activity measurements as a tool for demonstrating the possible cause of fish decay. Acta Biol. Szeged. 31, 9–12.
Olson, D.L. and Christensen, G.M. (1980) Effects of water pollutants and other chemicals on fish acetylcholinesterase in vitro. Environ. Res. 21, 327–35.
Readman, J.W., Liong Wee Kwong, L., Mee, L.D., Bartocci, J., Nilve, G., Rodriguez-Solano, J.A. and Gonzales-Farias, F. (1992) Persistent organophosphorous pesticides in tropical marine environments. Mar. Pollut. Bull. 24(8), 398–402.
Sprague, J.B. (1970) Measurement of pollutant toxicity to fish. II. Utilizing and applying bioassay results. Water Res. 4, 3–32.
Toutant, J.P. and Massoulié, J. (1988) Cholinesterases: tissue and cellular distribution of molecular forms and their physiological regulation. In V.P. Whittaker Handbook of experimental Pharmacology. 86, 167–224 Springer-Verlag, Berlin.
Wang, C. and Murphy, S.D. (1982) Kinetic analysis of species difference in acetylcholinesterase sensitivity to organophosphate insecticides. Toxicol. Appl. Pharmacol. 66, 409–19.
WHO (1986) IPCS. International Programme on Chemical Safety. Organophosphorous Insecticides, a General Introduction. Environmental Health Criteria 63, OMS, Geneva.
Zinkl, G.J., Shea, P.J., Nakamoto, R.J. and Callman, J. (1987) Brain cholinesterase activity of rainbow trout poisoned by carbaryl. Bull. Environ. Contam. Toxicol. 38, 29–35.
Zhu, K.Y. and Brindley, W.A. (1992) Molecular properties of acetylcholinesterase purified from Lygus hesperus knigh. Insect Biochem. Mol. Biol. 22(3), 253–60.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Bocquené, G., Bellanger, C., Cadiou, Y. et al. Joint action of combinations of pollutants on the acetylcholinesterase activity of several marine species. Ecotoxicology 4, 266–279 (1995). https://doi.org/10.1007/BF00116345
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00116345