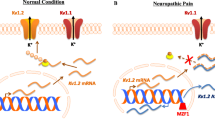
Abstract
RNA interference (RNAi) has become a powerful tool for modulating gene expression. While delivery of small interfering RNAs (siRNAs) has achieved silencing of pain-related genes in various animal models of nociception, delivery of short-hairpin RNA (shRNA) or artificial miRNA (miRNA) to dorsal root ganglia (DRG) has proven particularly challenging. This chapter describes a highly efficient method for in vivo gene silencing in sensory neurons using replication-defective vectors based on herpes simplex virus (HSV). This method can be utilised to obtain a better understanding of gene function, validate novel gene targets in drug discovery and potentially develop new RNAi-mediated approaches to achieve analgesia.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Lee RC, Feinbaum RL, Ambros V (1993) The C. elegans heterochronic gene lin-4 encodes small RNAs with antisense complementarity to lin-14. Cell 75(5):843–854
Lewis BP, Burge CB, Bartel DP (2005) Conserved seed pairing, often flanked by adenosines, indicates that thousands of human genes are microrna targets. Cell 120(1):15–20
Gregory RI, Yan K-P, Amuthan G, Chendrimada T, Doratotaj B, Cooch N, Shiekhattar R (2004) The microprocessor complex mediates the genesis of micrornas. Nature 432(7014):235–240
Lund E, Guttinger S, Calado A, Dahlberg JE, Kutay U (2004) Nuclear export of microrna precursors. Science 303(5654):95–98
Han J, Lee Y, Yeom K-H, Kim Y-K, Jin H, Kim VN (2004) The Drosha-DGCR8 complex in primary microrna processing. Genes Dev 18(24):3016–3027
Bernstein E, Caudy AA, Hammond SM, Hannon GJ (2001) Role for a bidentate ribonuclease in the initiation step of RNA interference. Nature 409(6818):363–366
Hutvagner G, Mclachlan J, Pasquinelli AE, Balint E, Tuschl T, Zamore PD (2001) A cellular function for the RNA-interference enzyme dicer in the maturation of the let-7 small temporal RNA. Science 293(5531):834–838
Khvorova A, Reynolds A, Jayasena SD (2003) Functional siRNAs and miRNAs exhibit strand bias. Cell 115(2):209–216
Fire A, Xu S, Montgomery MK, Kostas SA, Driver SE, Mello CC (1998) Potent and specific genetic interference by double-stranded RNA in Caenorhabditis elegans. Nature 391(6669):806–811
Jackson AL, Bartz SR, Schelter J, Kobayashi SV, Burchard J, Mao M, Li B, Cavet G, Linsley PS (2003) Expression profiling reveals off-target gene regulation by RNAi. Nat Biotech 21(6):635–637
Zeng Y, Wagner EJ, Cullen BR (2002) Both natural and designed micro RNAs can inhibit the expression of cognate mRNAs when expressed in human cells. Mol Cell 9(6):1327–1333
Zeng Y, Cullen BR (2003) Sequence requirements for micro RNA processing and function in human cells. RNA 9(1):112–123
Chung KH, Hart CC, Al-Bassam S, Avery A, Taylor J, Patel PD, Vojtek AB, Turner DL (2006) Polycistronic RNA polymerase II expression vectors for RNA interference based on BIC/miR-155. Nucleic Acids Res 34(7):e53
Silva JM, Li MZ, Chang K, Ge W, Golding MC, Rickles RJ, Siolas D, Hu G, Paddison PJ, Schlabach MR, Sheth N, Bradshaw J, Burchard J, Kulkarni A, Cavet G, Sachidanandam R, Mccombie WR, Cleary MA, Elledge SJ, Hannon GJ (2005) Second-generation shRNA libraries covering the mouse and human genomes. Nat Genet 37(11):1281–1288
Boudreau RL, Martins I, Davidson BL (2008) Artificial microRNAs as siRNA shuttles: improved safety as compared to shRNAs in vitro and in vivo. Mol Ther 17(1):169–175
Alvarez VA, Ridenour DA, Sabatini BL (2006) Retraction of synapses and dendritic spines induced by off-target effects of RNA interference. J Neurosci 26(30):7820–7825
Cao W, Hunter R, Strnatka D, Mcqueen CA, Erickson RP (2005) DNA constructs designed to produce short hairpin, interfering RNAs in transgenic mice sometimes show early lethality and an interferon response. J Appl Genet 46(2):217–225
Pebernard S, Iggo RD (2004) Determinants of interferon-stimulated gene induction by RNAi vectors. Differentiation 72:103–111
Bauer M, Kinkl N, Meixner A, Kremmer E, Riemenschneider M, Forstl H, Gasser T, Ueffing M (2008) Prevention of interferon-stimulated gene expression using microRNA-designed hairpins. Gene Ther 16(1):142–147
Mcbride JL, Boudreau RL, Harper SQ, Staber PD, Monteys AM, Martins IS, Gilmore BL, Burstein H, Peluso RW, Polisky B, Carter BJ, Davidson BL (2008) Artificial miRNAs mitigate shRNA-mediated toxicity in the brain: implications for the therapeutic development of RNAi. Proc Natl Acad Sci USA 105(15):5868–5873
Hutvágner G, Mj S, Cc M, Pd Z (2004) Sequence-specific inhibition of small RNA function. PLoS Biol 2(4):e98
Yi R, Doehle BP, Qin Y, Macara IG, Cullen BR (2005) Overexpression of exportin 5 enhances RNA interference mediated by short hairpin RNAs and microRNAs. RNA 11(2):220–226
Castanotto D, Sakurai K, Lingeman R, Li H, Shively L, Aagaard L, Soifer H, Gatignol A, Riggs A, Rossi JJ (2007) Combinatorial delivery of small interfering RNAs reduces RNAi efficacy by selective incorporation into RISC. Nucleic Acids Res 35(15):5154–5164
Grimm D, Streetz KL, Jopling CL, Storm TA, Pandey K, Davis CR, Marion P, Salazar F, Kay MA (2006) Fatality in mice due to oversaturation of cellular microRNA/short hairpin RNA pathways. Nature 441(7092):537–541
Dorn G, Patel S, Wotherspoon G, Hemmings-Mieszczak M, Barclay J, Natt FJC, Martin P, Bevan S, Fox A, Ganju P, Wishart W, Hall J (2004) SiRNA relieves chronic neuropathic pain. Nucleic Acids Res 32(5):e49
Luo MC, Zhang DQ, Ma SW, Huang YY, Shuster S, Porreca F, Lai J (2005) An efficient intrathecal delivery of small interfering RNA to the spinal cord and peripheral neurons. Mol Pain 1(1):29
Tan PH, Yang LC, Shih HC, Lan KC, Cheng JT (2004) Gene knockdown with intrathecal siRNA of NMDA receptor NR2B subunit reduces formalin-induced nociception in the rat. Gene Ther 12(1):59–66
Christoph T, Grünweller A, Mika J, Schäfer MKH, Wade EJ, Weihe E, Erdmann VA, Frank R, Gillen C, Kurreck J (2006) Silencing of vanilloid receptor TRPV1 by RNAi reduces neuropathic and visceral pain in vivo. Biochem Biophys Res Commun 350(1):238–243
Ralph GS, Radcliffe PA, Day DM, Carthy JM, Leroux MA, Lee DCP, Wong LF, Bilsland LG, Greensmith L, Kingsman SM, Mitrophanous KA, Mazarakis ND, Azzouz M (2005) Silencing mutant SOD1 using RNAi protects against neurodegeneration and extends survival in an ALS model. Nat Med 11(43):429–433
Raoul C, Abbas-Terki T, Bensadoun JC, Guillot S, Haase G, Szulc J, Henderson CE, Aebischer P (2005) Lentiviral-mediated silencing of SOD1 through RNA interference retards disease onset and progression in a mouse model of ALS. Nat Med 11(43):423–428
Harper SQ, Staber PD, He X, Eliason SL, Martins ISH, Mao Q, Yang L, Kotin RM, Paulson HL, Davidson BL (2005) RNA interference improves motor and neuropathological abnormalities in a huntington’s disease mouse model. Proc Natl Acad Sci USA 102(16):5820–5825
Singer O, Marr RA, Rockenstein E, Crews L, Coufal NG, Gage FH, Verma IM, Masliah E (2005) Targeting BACE1 with siRNAs ameliorates Alzheimer disease neuropathology in a transgenic model. Nat Neurosci 8(10):1343–1349
Xia H, Mao Q, Eliason SL, Harper SQ, Martins IH, Orr HT, Paulson HL, Yang L, Kotin RM, Davidson BL (2004) RNAi suppresses polyglutamine-induced neurodegeneration in a model of spinocerebellar ataxia. Nat Med 10(8):816–820
Palmer JA, Branston RH, Lilley CE, Robinson MJ, Groutsi F, Smith J, Latchman DS, Coffin RS (2000) Development and optimization of herpes simplex virus vectors for multiple long-term gene delivery to the peripheral nervous system. J Virol 74(12):5604–5618
Lilley CE, Groutsi F, Han Z, Palmer JA, Anderson PN, Latchman DS, Coffin RS (2001) Multiple immediate-early gene-deficient herpes simplex virus vectors allowing efficient gene delivery to neurons in culture and widespread gene delivery to the central nervous system in vivo. J Virol 75(9):4343–4356
Anesti AM, Peeters PJ, Royaux I, Coffin RS (2008) Efficient delivery of RNA interference to peripheral neurons in vivo using herpes simplex virus. Nucleic Acids Res 36(14):e86
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2010 Springer Science+Business Media, LLC
About this protocol
Cite this protocol
Anesti, AM. (2010). Delivery of RNA Interference to Peripheral Neurons In Vivo Using Herpes Simplex Virus. In: Szallasi, A. (eds) Analgesia. Methods in Molecular Biology, vol 617. Humana Press, Totowa, NJ. https://doi.org/10.1007/978-1-60327-323-7_26
Download citation
DOI: https://doi.org/10.1007/978-1-60327-323-7_26
Published:
Publisher Name: Humana Press, Totowa, NJ
Print ISBN: 978-1-60327-322-0
Online ISBN: 978-1-60327-323-7
eBook Packages: Springer Protocols