Abstract
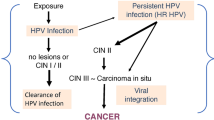
Cervical cancer is the third most common type of cancer in women in the United States and is the leading cause of cancer deaths in women in third world countries (1,2). Although the pathogenesis of the disease is incompletely understood, human papillomavirus (HPV) is present in over 90% of all high grade cervical lesions and is strongly implicated in the process of cancer development. A common feature of these tumors is expression of the E6 and E7 viral reading frames (3,4,5). These viral genes encode factors, E6 and E7, that interact with the p53 and pRB tumor suppressor genes (6,7,8,9,10). E6 promotes the degradation of p53 via a ubiquitin-dependent pathway (10,11) and E7 interferes with pRB function (8,9,10). Expression of E6 and E7 is sufficient for immortalization of cultured epidermal keratinocytes and cultured cervical epithelial cells (12,13,14,15).
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Preview
Unable to display preview. Download preview PDF.
Similar content being viewed by others
References
P.J. Disaia, and W.T. Creasman, Preinvasive disease of the cervix, in: “Clinical Gynecologic Oncology,” P.J. Disaia, and W.T. Creasman, eds. Mosby Year Book, St Louis (1993) pp. 1–36.
American Cancer Society: Cancer Facts and Figures. American Cancer Society, Atlanta (1992).
T.R. Broker, Structure and genetic expression of papillomaviruses, Obstet. Gynecol. Clin. North. Am., 14: 329 (1987).
H. zur Hausen, Human papillomaviruses and their possible role in squamous cell carcinomas, Curr. Top. Microbiol. Immunol., 78: 1 (1977).
L. Gissmann, M. Boshart, M. Durst, H. Ikenberg, D. Wagner, and H. zur Hausen, Presence of human papillomavirus in genital tumors, J. Invest. Dermatol., 83: 26s (1984).
M.S. Lechner, D.H. Mack, A.B. Finkle, T. Crook, K.H. Vousden, and L A Laimins, Human papillomavirus E6 proteins bind p53 in vivo and abrogate p53-mediated repression of transcription, EMBO J., 11: 3045 (1992).
V. Band, J.A. De Caprio, L. Delmolino, V. Kulesa, and R. Sager, Loss of p53 protein in human papillomavirus type 16 E6- immortalized human mammary epithelial cells, J. Virol., 65: 6671 (1991).
P.S. Huang, D.R. Patrick, G. Edwards, P.J. Goodhart, H.E. Huber, L. Miles, V.M. Garsky, A. Oliff, and D.C. Heimbrook, Protein domains governing interactions between E2F, the retinoblastoma gene product, and human papillomavirus type 16 E7 protein, Mol. Cell. Biol. 13: 953 (1993).
D.V. Heck, C.L. Yee, P.M. Howley, and K. Munger, Efficiency of binding the retinoblastoma protein correlates with the transforming capacity of the E7 oncoproteins of the human papillomaviruses, Proc. Natl. Acad. Sci. USA. 89: 4442 (1992).
K. Munger, M. Scheffner, J.M. Huibregtse, and P.M. Howley, Interactions of HPV E6 and E7 oncoproteins with tumour suppressor gene products. Cancer. Surv., 12: 197 (1992).
J.M. Huibregtse, M. Scheffner, and P.M. Howley, A cellular protein mediates association of p53 with the E6 oncoprotein of human papillomavirus types 16 or 18, EMBO J., 10: 4129 (1991).
P. Hawley-Nelson, K.H. Vousden, N.L. Hubbert, D.R. Lowy, and J.T. Schiller, HPV 16 E6 and E7 proteins cooperate to immortalize human foreskin keratinocytes, EMBO J., 8: 3905 (1989).
L. Pirisi, S. Yasumoto, M. Feller, J. Domger, and J.A. DiPaolo, Transformation of human fibroblasts and keratinocytes with human papillomavirus type 16 DNA, J. Viral., 61: 1061 (1987).
M.S. Barbosa, and R. Schlegel, The E6 and E7 genes of HPV-18 are sufficient for inducing two-stage in vitro transformation of human keratinocytes, Oncogene, 4: 1529 (1989).
C.D. Woodworth, J. Doniger, and J.A. DiPaolo, Immortalization of human foreskin keratinocytes by various human papillomavirus DNAs corresponds to their association with cervical carcinoma, J. Virol.,. 63: 159 (1989).
A.P. Cullen, R. Reid, M. Campion, and A.T. Lorincz, Analysis of the physical state of different human papillomavirus DNAs in intraepithelial and invasive cervical neoplasm, J. Virol., 65: 606 (1991).
C.M. Chiang, G. Dong, T.R. Broker, and L.T. Chow, Control of human papillomavirus type 11 origin of replication by the E2 family of transcription regulatory proteins, J. Virol., 66: 5224 (1992).
C.M. Chiang, M. Ustav, A. Stenlund, T.F. Ho, T.R. Broker, and L.T. Chow, Viral El and E2 proteins support replication of homologous and heterologous papillomaviral origins, Proc. Natl. Acad. Sci. U S A., 89: 5799 (1992).
H. Romanczuk, and P.M. Howley, Disruption of either the El or the E2 regulatory gene of human papillomavirus type 16 increases viral immortalization capacity, Proc. Natl. Acad. Sci. USA., 89: 3159 (1992).
B.C. Sang, and M.S. Barbosa, Increased E6/E7 transcription in HPV 18-immortalized human keratinocytes results from inactivation of E2 and additional cellular events, Virology, 189: 448 (1992).
R. Sousa, N. Dostatni, and M. Yaniv, Control of papillomavirus gene expression, Biochim. Biophys. Acta, 1032: 19 (1990).
S.L. Romnay, P.R. Palan, and C. Duttagupta, S. Wassertheil-Smolter, J. Wylie, G. Miller, N.S. Slagle, and D. Lucido, Retinoids and the prevention of cervical dysplasia, Am. J. Obst. Gyn., 141: 890 (1981).
W.J. Winkelstein, E.J. Shillitoe, R. Brand, and K.K. Johnson, Further comments on cancer of the uterine cervix, smoking, and herpesvirus infection, Am. J. Epidemiol., 119: 1 (1984).
H.Y. Ngan, R.J. Collins, K.Y. Wong, A. Cheung, C.F. Lai, and Y.T. Liu, Cervical human papilloma virus infection of women attending social hygiene clinics in Hong Kong, Int. J. Gynaecol. Obstet., 41: 75 (1993).
S.M. Lippman, B.S. Glisson, J.J. Kavanagh, R. Lotan, W.K. Hong, M. Paredes Espinoza, W.N. Hittelman, E.E. Holdener, and I.H. Krakoff, Retinoic acid and interferon combination studies in human cancer, Eur. J. Cancer, 29A Suppl. 5: S9 (1993).
S.M. Lippman, J.J. Kavanagh, M. Paredes Espinoza, F. Delgadillo Madrueno, P. Paredes Casillas, W.K. Hong, G. Massimini, E.E. Holdener, and I.H. Krakoff, 13-cis-retinoic acid plus interferon-alpha 2a in locally advanced squamous cell carcinoma of the cervix, J. Natl. Cancer Inst., 85: 499 (1993).
S.M. Lippman, and W.K. Hong, 13-cis-retinoic acid and cancer chemoprevention, Monogr. Natl. Cancer Inst., 13: 111 (1992).
C. Agarwal, E.A. Rorke, J.C. Irwin, and R.L. Eckert, Immortalization by human papillomavirus type 16 alters retinoid regulation of human ectocervical epithelial cell differentiation, Cancer Res., 51: 3982 (1991).
C. Agarwal, J.R. Hembree, E.A. Rorke, and R.L. Eckert, Interferon and retinoic acid suppress the growth of human papillomavirus type 16 immortalized cervical epithelial cells, but only interferon suppresses the level of the human papillomavirus transforming oncogenes, Cancer Res., 54: 2108 (1994).
L. Pirisi, A. Batova, G.R. Jenkins, J.R. Hodam, and K.E. Creek, Increased sensitivity of human keratinocytes immortalized by human papillomavirus type 16 DNA to growth control by retinoids, Cancer Res., 52: 187 (1992).
G.I. Gorodeski, R.L. Eckert, W.H. Utian, L. Sheean, and E.A. Rorke, Retinoids, sex steroids and glucocorticoids regulate ectocervical cell envelope formation but not the level of the envelope precursor, involucrin, Differentiation 42: 75 (1989).
G.I. Gorodeski, R.L. Eckert, W.H. Utian and E.A. Rorke, Maintenance of in vivo-like keratin expression, sex steroid responsiveness and estrogen receptor expression in cultured human ectocervical epithelial cells, Endocrinology 126: 399 (1990).
G.I. Gorodeski, R.L. Eckert, W.H. Utian, L. Sheean and E.A. Rorke, Cultured human ectocervical epithelial cell differentiation is regulated by the combined direct actions of sex steroids, glucocorticoids and retinoids, J. Clin. Endocrinol. Metab. 70: 1624 (1990).
C.K. Choo, E.A. Rorke and R.L. Eckert, Calcium regulates the differentiation of human papillomavirus type 16 immortalized ectocervical epithelial cells, but not the expression of the papillomavirus E6 and E7 oncogenes, Exper. Cell Res. 208: 161 (1993).
C.K. Choo, E.A. Rorke, and R.L. Eckert, Differentiation-dependent constitutive expression of the human papillomavirus type 16 E6 and E7 oncogenes in CaSki cervical tumor cells, J. Gen. Virol. 75: 1139 (1994).
N. Sizemore, and E.A. Rorke, Retinoid regulation of human ectocervical epithelial cell transglutaminase activity and keratin gene expression, Differentiation 54: 219 (1993).
L. Kasturi, N. Sizemore, R.L. Eckert, K. Martin, and E.A. Rorke, Calcium modulates cornified envelope formation, involucrin content, and transglutaminase activity in cultured human ectocervical epithelial cells, Exp. Cell. Res. 205: 84 (1993).
S. Andreatta-van Leyen, J.R. Hembree, and R.L. Eckert, Regulation of IGF-I binding protein 3 levels by epidermal growth factor and retinoic acid in cervical epithelial cells, J Cell. Physiol. 160: 265 (1994).
J. Hembree, C. Agarwal and R.L. Eckert, Epidermal growth factor suppresses insulin-like growth factor binding protein-3 levels in human papillomavirus type 16-immortalized cervical epithelial cells and thereby potentiates the effects of IGF-I, Cancer Res. 54: 3160 (1994).
N. Sizemore, and E.A. Rorke, Human papillomavirus 16 immortalization of normal human ectocervical epithelial cells alters retinoic acid regulation of cell growth and epidermal growth factor receptor expression, Cancer Res. 53: 4511 (1993).
C.K. Choo, E.A. Rorke, and R.L. Eckert, Human papillomavirus E6 and E7 oncogene expression is not correlated with cell proliferation or c-myc RNA expression in HPV 16-immortalized human cervical epithelial cells treated with retinoids, Cancer Res. (submitted).
R.A. Pattillo, R.O. Hussa, M.T. Story, A.C. Ruckert, M.R. Shalaby, and R.F. Mattingly, Tumor antigen and human chorionic gonadotropin in CaSki cells: a new epidermoid cervical cancer cell line, Science 196: 1456 (1977).
B.M. Gilfix, and R.L. Eckert, Coordinate control by vitamin A of keratin gene expression in human keratinocytes, J. Biol. Chem. 260: 14026 (1985).
R.L. Eckert and H. Green, Cloning of cDNAs specifying vitamin A-responsive human keratins, Proc. Natl. Acad. Sci. USA 81: 4321 (1984).
M. Athar, R. Agarwal, Z.Y. Wang, J.R. Lloyd, D.R. Bickers and H. Mukhtar, All-trans-retinoic acid protects against conversion of chemically induced and ultraviolet B radiation induced skin papillomas to carcinomas, Carcinogenesis 12: 2325 (1991).
N. Sizemore, L. Kasturi, G. Gorodeski R.L. Eckert, A.M. Jetten and E.A. Rorke, Retinoids regulate human ectocervical epithelial cell transglutaminase activity and keratin gene expression, Differentiation 54: 219 (1993).
R.L. Eckert, Structure, function, and differentiation of the keratinocyte, Physiol. Rev., 69: 1316 (1989).
W.S. Cohick and D.R. Clemmons, Regulation of insulin-like growth factor binding protein synthesis and secretion in a bovine epithelial cell line, Endocrinology, 129: 1347 (1991).
D.P. DeLeon, B. Bakker, D.M. Wilson, R.L. Hintz, and R.G. Rosenfeld, Demonstration of insulin-like growth factor (IGF-I and -II) receptors and binding proteins in human breast cancer cell lines, Biochem. Biophys. Res. Comun., 152: 398 (1988).
R.C. Baxter, and J.L. Martin, Binding proteins for insulin-like growth factors: structure, regulation and function, Prog. Growth Factor Res. 1: 49 (1989).
N. Darwiche, G. Celli, L. Sly, F. Lancillotti, and L.M. De Luca, Retinoid status controls the appearance of reserve cells and keratin expression in mouse cervical epithelium, Cancer Res., 53: 2287 (1993).
R. Moll, W.W. Franke, D.L. Schiller, B. Geiger, and R. Krepler, The catalog of human cytokeratins: patterns of expression in normal epithelia, tumors and cultured cells, Cell 31: 11 (1982).
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 1995 Springer Science+Business Media New York
About this chapter
Cite this chapter
Eckert, R.L. et al. (1995). Human Cervical Cancer. In: Diet and Cancer. Advances in Experimental Medicine and Biology, vol 354. Springer, Boston, MA. https://doi.org/10.1007/978-1-4899-0949-7_3
Download citation
DOI: https://doi.org/10.1007/978-1-4899-0949-7_3
Publisher Name: Springer, Boston, MA
Print ISBN: 978-1-4899-0951-0
Online ISBN: 978-1-4899-0949-7
eBook Packages: Springer Book Archive