Abstract
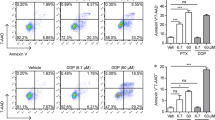
Ajoene, a garlic-derived organosulfur compound, exerts anti-tumorigenic effect against various cancers. However, little is known about the biological effect of ajoene on lung adenocarcinoma, an aggressive malignancy with dismal prognosis. We investigated the biological effect of ajoene on lung adenocarcinoma and the underlying pathway. Lung adenocarcinoma cells A549, NCI-H1373, and NCI-H1395, along with the noncancerous lung bronchus cells BEAS-2B, were used. MTT test showed that ajoene (25 μM) reduces viability of lung adenocarcinoma cells but not the noncancerous BEAS-2B cells. Bromodeoxyuridine incorporation assay revealed that ajoene inhibits proliferation of lung adenocarcinoma cells. Treatment of lung adenocarcinoma cells with ajoene enhances apoptosis and ROS generation in a time- and dose-dependent fashion. Abrogation of caspase activation by zVAD-fmk completely prevents the ajoene-induced apoptosis; whereas block of ROS generation by N-acetylcysteine partly abolishes the ajoene-induced apoptosis. ROS-mediated induction of apoptosis contributes partially to the anti-tumorigenic property of ajoene observed, a phenomenon also confirmed by xenograft tumor study. Mitogen activated protein kinases (MAPKs), pivots of ROS-mediated signaling pathway, are activated upon ajoene treatment; Jun-N-terminal kinase (JNK)/p38 activations are required for signaling pathway underlying the ajoene-induced apoptosis. Our results suggest that ROS-mediated activation of JNK/p38 contributes partially to the pro-apoptotic action of ajoene on cells of lung adenocarcinoma. Ajoene may be a promising chemotherapeutic agent for lung adenocarcinoma.







Similar content being viewed by others
References
Leng S, Liu Y, Weissfeld JL, Thomas CL, Han Y, Picchi MA, et al. 15q12 variants, sputum gene promoter hypermethylation, and lung cancer risk: a GWAS in smokers. J Natl Cancer Inst. 2015;107.
Ding L, Getz G, Wheeler DA, Mardis ER, McLellan MD, Cibulskis K, et al. Somatic mutations affect key pathways in lung adenocarcinoma. Nature. 2008;455:1069–75.
Wensing KU, Ciarimboli G. Saving ears and kidneys from cisplatin. Anticancer Res. 2013;33:4183–8.
Londhe VP, Gavasane AT, Nipate SS, Bandawane DD, Chaudhari PD. Role of garlic (Allium sativum) in various diseases—an overview. JPRO. 2011;1:129–34.
Wali IE, Awad AMR. Combined activity of garlic and nitrofurantoin against Escherichia coli and enterococcus species recovered from urinary tract infections. AJMR. 2014;8:644–51.
Miroddi M, Calapai F, Calapai G. Potential beneficial effects of garlic in oncohematology. Mini Rev Med Chem. 2011;11:461–72.
Apitz-Castro R, Badimon JJ, Badimon L. Effect of ajoene, the major antiplatelet compound from garlic, on platelet thrombus formation. Thromb Res. 1992;68:145–55.
Apitz-Castro R, Badimon JJ, Badimon L. A garlic derivative, ajoene, inhibits platelet deposition on severely damaged vessel wall in an in vivo porcine experimental model. Thromb Res. 1994;75:243–9.
Apitz-Castro R, Escalante J, Vargas R, Jain MK. Ajoene, the antiplatelet principle of garlic, synergistically potentiates the antiaggregatory action of prostacyclin, forskolin, indomethacin and dypiridamole on human platelets. Thromb Res. 1986;42:303–11.
Gebhardt R, Beck H, Wagner KG. Inhibition of cholesterol biosynthesis by allicin and ajoene in rat hepatocytes and HepG2 cells. Biochim Biophys Acta. 1994;1213:57–62.
Kaschula CH, Hunter R, Stellenboom N, Caira MR, Winks S, Ogunleye T, et al. Structure-activity studies on the anti-proliferation activity of ajoene analogues in WHCO1 oesophageal cancer cells. Eur J Med Chem. 2012;50:236–54.
Taylor P, Noriega R, Farah C, Abad MJ, Arsenak M, Apitz R. Ajoene inhibits both primary tumor growth and metastasis of B16/BL6 melanoma cells in C57BL/6 mice. Cancer Lett. 2006;239:298–304.
Dirsch VM, Antlsperger DS, Hentze H, Vollmar AM. Ajoene, an experimental anti-leukemic drug: mechanism of cell death. Leukemia. 2002;16:74–83.
Dirsch VM, Gerbes AL, Vollmar AM. Ajoene, a compound of garlic, induces apoptosis in human promyeloleukemic cells, accompanied by generation of reactive oxygen species and activation of nuclear factor kappaB. Mol Pharmacol. 1998;53:402–7.
Dirsch VM, Vollmar AM. Ajoene, a natural product with non-steroidal anti-inflammatory drug (NSAID)-like properties? Biochem Pharmacol. 2001;61:587–93.
da Lee Y, Li H, Lim HJ, Lee HJ, Jeon R, Ryu JH. Anti-inflammatory activity of sulfur-containing compounds from garlic. J Med Food. 2012;15:992–9.
Kay HY, Won Yang J, Kim TH, Lee da Y, Kang B, Ryu JH, et al. Ajoene, a stable garlic by-product, has an antioxidant effect through Nrf2-mediated glutamate-cysteine ligase induction in HepG2 cells and primary hepatocytes. J Nutr. 2010;140:1211–9.
Bergethon K, Shaw AT, Ou SH, Katayama R, Lovly CM, McDonald NT, et al. ROS1 rearrangements define a unique molecular class of lung cancers. J Clin Oncol. 2012;30:863–70.
Hanly L, Figueredo R, Rieder MJ, Koropatnick J, Koren G. The effects of n-acetylcysteine on ifosfamide efficacy in a mouse xenograft model. Anticancer Res. 2012;32:3791–8.
Carrasco RA, Stamm NB, Marcusson E, Sandusky G, Iversen P, Patel BK. Antisense inhibition of survivin expression as a cancer therapeutic. Mol Cancer Ther. 2011;10:221–32.
Ledezma E, Apitz-Castro R, Cardier J. Apoptotic and anti-adhesion effect of ajoene, a garlic derived compound, on the murine melanoma B16F10 cells: possible role of caspase-3 and the alpha(4)beta(1) integrin. Cancer Lett. 2004;206:35–41.
Tilli CM, Stavast-Kooy AJ, Vuerstaek JD, Thissen MR, Krekels GA, Ramaekers FC, et al. The garlic-derived organosulfur component ajoene decreases basal cell carcinoma tumor size by inducing apoptosis. Arch Dermatol Res. 2003;295:117–23.
Yang JY, Della-Fera MA, Nelson-Dooley C, Baile CA. Molecular mechanisms of apoptosis induced by ajoene in 3T3-L1 adipocytes. Obesity (Silver Spring). 2006;14:388–97.
Jacobson MD, Raff MC. Programmed cell death and Bcl-2 protection in very low oxygen. Nature. 1995;374:814–6.
Ray PD, Huang BW, Tsuji Y. Reactive oxygen species (ROS) homeostasis and redox regulation in cellular signaling. Cell Signal. 2012;24:981–90.
Ichijo H, Nishida E, Irie K, ten Dijke P, Saitoh M, Moriguchi T, et al. Induction of apoptosis by ASK1, a mammalian MAPKKK that activates SAPK/JNK and p38 signaling pathways. Science. 1997;275:90–4.
Jung Y, Park H, Zhao HY, Jeon R, Ryu JH, Kim WY. Systemic approaches identify a garlic-derived chemical, Z-ajoene, as a glioblastoma multiforme cancer stem cell-specific targeting agent. Mol Cells. 2014;37:547–53.
Scharfenberg K, Wagner R, Wagner KG. The cytotoxic effect of ajoene, a natural product from garlic, investigated with different cell lines. Cancer Lett. 1990;53:103–8.
Chakraborti T, Das S, Mondal M, Roychoudhury S, Chakraborti S. Oxidant, mitochondria and calcium: an overview. Cell Signal. 1999;11:77–85.
McConkey DJ, Orrenius S. The role of calcium in the regulation of apoptosis. Biochem Biophys Res Commun. 1997;239:357–66.
Takadera T, Ohyashiki T. Apoptotic cell death and caspase 3 (CPP32) activation induced by calcium ionophore at low concentrations and their prevention by nerve growth factor in PC12 cells. Eur J Biochem. 1997;249:8–12.
Ark EK, Kwon KB, Park KI, Park BH, Jhee EC. Role of Ca(2+) in diallyl disulfide-induced apoptotic cell death of HCT-15 cells. Exp Mol Med. 2002;34:250–7.
Chen WC, Hsu SS, Chou CT, Kuo CC, Huang JK, Fang YC, et al. Effect of diallyl disulfide on Ca2+ movement and viability in PC3 human prostate cancer cells. Toxicol In Vitro. 2011;25:636–43.
Juin P, Pelletier M, Oliver L, Tremblais K, Gregoire M, Meflah K, et al. Induction of a caspase-3-like activity by calcium in normal cytosolic extracts triggers nuclear apoptosis in a cell-free system. J Biol Chem. 1998;273:17559–64.
Moran J, Itoh T, Reddy UR, Chen M, Alnemri ES, Pleasure D. Caspase-3 expression by cerebellar granule neurons is regulated by calcium and cyclic AMP. J Neurochem. 1999;73:568–77.
Shen HM, Dong SY, Ong CN. Critical role of calcium overloading in cadmium-induced apoptosis in mouse thymocytes. Toxicol Appl Pharmacol. 2001;171:12–9.
Shen YH, Godlewski J, Zhu J, Sathyanarayana P, Leaner V, Birrer MJ, et al. Cross-talk between JNK/SAPK and ERK/MAPK pathways: sustained activation of JNK blocks ERK activation by mitogenic factors. J Biol Chem. 2003;278:26715–21.
Avisetti DR, Babu KS, Kalivendi SV. Activation of p38/JNK pathway is responsible for embelin induced apoptosis in lung cancer cells: transitional role of reactive oxygen species. PLoS One. 2014;9, e87050.
Tsujimoto A, Takemura G, Mikami A, Aoyama T, Ohno T, Maruyama R, et al. A therapeutic dose of the lipophilic statin pitavastatin enhances oxidant-induced apoptosis in human vascular smooth muscle cells. J Cardiovasc Pharmacol. 2006;48:160–5.
Kim KN, Ham YM, Moon JY, Kim MJ, Jung YH, Jeon YJ, et al. Acanthoic acid induces cell apoptosis through activation of the p38 MAPK pathway in HL-60 human promyelocytic leukaemia. Food Chem. 2012;135:2112–7.
Hyun MS, Hur JM, Mun YJ, Kim D, Woo WH. BBR induces apoptosis in HepG2 cell through an Akt-ASK1-ROS-p38MAPKs-linked cascade. J Cell Biochem. 2010;109:329–38.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
None
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(PDF 1082 kb)
Rights and permissions
About this article
Cite this article
Wang, Y., Sun, Z., Chen, S. et al. ROS-mediated activation of JNK/p38 contributes partially to the pro-apoptotic effect of ajoene on cells of lung adenocarcinoma. Tumor Biol. 37, 3727–3738 (2016). https://doi.org/10.1007/s13277-015-4181-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13277-015-4181-9