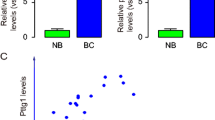
Abstract
Increased expression of pituitary tumor-transforming gene 1 (PTTG1) occurs during mitosis-related sister chromatid segregation, and characterizes various tumor cells, including prostate cancer. Whereas the mechanism remains unclarified. Here, the PTTG1 levels in a prostate cancer cell line, PC3, were modulated by the expression of PTTG1 transgene or shRNA, showing that the PTTG1 levels affected the proliferation of prostate cancer cells, in vitro and in vivo. Moreover, a significant decrease in mothers against decapentaplegic homolog 3 (SMAD3), a key component of transforming growth factor β (TGFβ) signaling pathway, was induced by PTTG1 overexpression. Since SMAD3 is a ubiquitous cell-cycle inhibitor, our data suggest that PTTG1 may promote the proliferation of prostate cancer cells by inhibiting SMAD3-mediated TGFβ signaling. To identify a causal link, we expressed SMAD3 in PTTG1-overexpressing PC3 cells and found that SMAD3 expression inhibited the augmented cancer cell proliferation by PTTG1overexpression. Furthermore, SMAD3 inhibition by short hairpin RNA (ShRNA) completely rescued the cancer cell proliferation in PTTG1 ShRNA-treated PC3 cells. Taken together, our data suggest that PTTG1 promotes the proliferation of prostate cancer cells via the inhibition of SMAD3. SMAD3 thus appears to be a novel therapeutic target for suppressing the growth of prostate cancer.




Similar content being viewed by others
References
Zhang X et al. Structure, expression, and function of human pituitary tumor-transforming gene (PTTG). Mol Endocrinol. 1999;13(1):156–66.
Yu R et al. Pituitary tumor transforming gene (PTTG) regulates placental JEG-3 cell division and survival: evidence from live cell imaging. Mol Endocrinol. 2000;14(8):1137–46.
Panguluri SK, Yeakel C, Kakar SS. PTTG: an important target gene for ovarian cancer therapy. J Ovarian Res. 2008;1(1):6.
El-Naggar SM, Malik MT, Kakar SS. Small interfering RNA against PTTG: a novel therapy for ovarian cancer. Int J Oncol. 2007;31(1):137–43.
Chen G et al. Inhibitory effects of anti-sense PTTG on malignant phenotype of human ovarian carcinoma cell line SK-OV-3. J Huazhong Univ Sci Technolog Med Sci. 2004;24(4):369–72.
Zhang J. et al. Overexpression of pituitary tumor transforming gene (PTTG) is associated with tumor progression and poor prognosis in patients with esophageal squamous cell carcinoma. Acta Histochem 2013
Yan S et al. PTTG overexpression promotes lymph node metastasis in human esophageal squamous cell carcinoma. Cancer Res. 2009;69(8):3283–90.
Zhou C et al. Overexpression of human pituitary tumor transforming gene (hPTTG), is regulated by beta-catenin /TCF pathway in human esophageal squamous cell carcinoma. Int J Cancer. 2005;113(6):891–8.
Shibata Y et al. Expression of PTTG (pituitary tumor transforming gene) in esophageal cancer. Jpn J Clin Oncol. 2002;32(7):233–7.
Zhang ML, Lu S, Zheng SS. Epigenetic changes of pituitary tumor-derived transforming gene 1 in pancreatic cancer. Hepatobiliary Pancreat Dis Int. 2008;7(3):313–7.
Ai J et al. Identification of over-expressed genes in human renal cell carcinoma by combining suppression subtractive hybridization and cDNA library array. Sci China C Life Sci. 2004;47(2):148–57.
Dominguez A et al. hpttg, a human homologue of rat pttg, is overexpressed in hematopoietic neoplasms. Evidence for a transcriptional activation function of hPTTG. Oncogene. 1998;17(17):2187–93.
Zhou C et al. PTTG acts as a STAT3 target gene for colorectal cancer cell growth and motility. Oncogene. 2014;33(7):851–61.
Kim DS et al. Securin induces genetic instability in colorectal cancer by inhibiting double-stranded DNA repair activity. Carcinogenesis. 2007;28(3):749–59.
Huang SQ et al. RNAi-mediated knockdown of pituitary tumor- transforming gene-1 (PTTG1) suppresses the proliferation and invasive potential of PC3 human prostate cancer cells. Braz J Med Biol Res. 2012;45(11):995–1001.
Massague J. TGFbeta in cancer. Cell. 2008;134(2):215–30.
Shi Y, Massague J. Mechanisms of TGF-beta signaling from cell membrane to the nucleus. Cell. 2003;113(6):685–700.
Kretzschmar M, Massague J. SMADs: mediators and regulators of TGF-beta signaling. Curr Opin Genet Dev. 1998;8(1):103–11.
Massague J, Chen YG. Controlling TGF-beta signaling. Genes Dev. 2000;14(6):627–44.
Massague J. TGFbeta signalling in context. Nat Rev Mol Cell Biol. 2012;13(10):616–30.
Pavese J, Ogden IM, Bergan RC. An orthotopic murine model of human prostate cancer metastasis. J Vis Exp. 2013;79:e50873.
Acknowledgments
This work was supported by the internal funding of the Third Military Medical University.
Conflict of interest
None
Author’s contributions
Study concept, funding, and manuscript—SH; acquisition, analysis, and interpretation of data—SH, QL, LL, and DX.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Huang, S., Liao, Q., Li, L. et al. PTTG1 inhibits SMAD3 in prostate cancer cells to promote their proliferation. Tumor Biol. 35, 6265–6270 (2014). https://doi.org/10.1007/s13277-014-1818-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13277-014-1818-z