Abstract
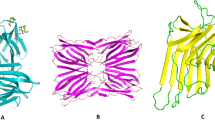
A mannose-binding lectin (Narcissus tazetta lectin [NTL]) with potent antiviral activity was isolated and purified from the bulbs of the Chinese daffodil Narcissus tazetta var. chinensis, using ion exchange chromatography on diethylaminoethyl (DEAE)-cellulose, affinity chromatography on mannose-agarose and fast protein liquid chromatography (FPLC)-gel filtration on Superose 12. The purified lectin was shown to have an apparent molecular mass of 26 kDa by gel filtration and 13 kDa by SDS-PAGE, indicating that it is probably a dimer with two identical subunits. The cDNA-derived amino acid sequence of NTL as determined by molecular cloning also reveals that NTL protein contains a mature polypeptide consisting of 105 amino acids and a C-terminal peptide extension. Three-dimensional modelling study demonstrated that the NTL primary polypeptide contains three subdomains, each with a conserved mannose-binding site. It shows a high homology of about 60%–80% similarity with the existing monocot mannose-binding lectins. NTL could significantly inhibit plaque formation by the human respiratory syncytial virus (RSV) with an IC50 of 2.30 µg/ml and exhibit strong antiviral properties against influenza A (H1N1, H3N2, H5N1) and influenza B viruses with IC50 values ranging from 0.20 µg/ml to 1.33 µg/ml in a dose-dependent manner. It is worth noting that the modes of antiviral action of NTL against RSV and influenza A virus are significantly different. NTL is effective in the inhibition of RSV during the whole viral infection cycle, but the antiviral activity of NTL is mainly expressed at the early stage of the viral cycle of influenza A (H1N1) virus. NTL with a high selective index (SI=CC50/IC50≥141) resulting from its potent antiviral activity and low cytotoxicity demonstrates a potential for biotechnological development as an antiviral agent.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Abbreviations
- 3D:
-
three-dimensional
- ATCC:
-
American Type Culture Collection
- CC:
-
cytotoxic concentration
- CoV:
-
coronavirus
- CPE:
-
cytopathic effect
- DEAE:
-
diethylaminoethyl
- DMSO:
-
dimethyl sulphoxide
- EMEM:
-
Eagle minimum essential medium
- FBS:
-
foetal bovine serum
- FPLC:
-
fast protein liquid chromatography
- GNA:
-
Galanthus nivalis agglutinin
- HA:
-
haemagglutination
- HCMV:
-
human cytomegalovirus
- HCV:
-
hepatitis C virus
- HEp2:
-
human larynx epidermoid carcinoma cell line
- HHA:
-
Hippeastrum hybrid agglutinin
- HIV:
-
human immunodeficiency virus
- HSV:
-
herpes simplex virus
- IC:
-
inhibitory concentration
- IFN:
-
interferon
- MDCK:
-
Madin-Darby canine kidney cell line
- MOI:
-
multiplicity of infection
- NTL:
-
Narcissus tazetta lectin
- OD:
-
optical density
- PBS:
-
phosphate-buffered saline
- RSV:
-
respiratory syncytial virus
- SCID:
-
severe combined immunodeficiency
- SDS:
-
sodium dodecyl sulphate
- SI:
-
selective index
- TGF:
-
transforming growth factor
- TPCK:
-
tolylsulphonyl phenylalanyl chloromethyl ketone
- UDA:
-
Urtica dioica agglutinin
References
Balzarini J 2006 Inhibition of HIV entry by carbohydrate-binding proteins; Antivir. Res. 71 237–247
Balzarini J 2007a Targeting the glycans of glycoproteins: a novel paradigm for antiviral therapy; Nat. Rev. Microbiol. 5 583–597
Balzarini J 2007b Carbohydrate-binding agents: a potential future cornerstone for the chemotherapy of enveloped viruses?; Antivir. Chem. Chemother. 18 1–11
Balzarini J, Neyts J, Schols D, Hosoya M, Van Damme E J M, Peumans W J and De Clercq E 1992 The mannose-specific plant lectins from Cymbidium hybrid and Epipactis helleborine and the (N-acetylglucosamine)n-specific plant lectin from Urtica dioica are potent and selective inhibitors of human immunodeficiency virus and cytomegalovirus replication in vitro; Antivir. Res. 18 191–207
Balzarini J, Schols D, Neyts J, Van Damme E J M, Peumans W J and De Clercq E 1991 Alpha-(1-3)- and alpha-(1-6)-D-mannose-specific plant lectins are markedly inhibitory to human immunodeficiency virus and cytomegalovirus infections in vitro; Antimicrob. Agents Chemother. 35 410–416
Barre A, Van Damme E J M, Peumans W J and Rouge P 1996 Structure-function relationship of monocot mannose-binding lectins; Plant Physiol. 112 1531–1540
Barre A, Bourne Y, Van Damme, E J M, Peumans W J and Rougé P 2001 Mannose-binding plant lectins: different structural scaffolds for a common sugar-recognition process; Biochimie 83 645–651
Bertaux C, Daelemans D, Meertens L, Cormier E G, Reinus J F, Peumans W J, Van Damme E J M, Igarashi Y, Oki T, Schols D, Dragic T and Balzarini J 2007 Entry of hepatitis C virus and human immunodeficiency virus is selectively inhibited by carbohydratebinding agents but not by polyanions; Virology 366 40–50
Feldman S A, Crim R L, Audet S A and Beeler J A 2001 Human respiratory syncytial virus surface glycoproteins F, G and SH form an oligomeric complex; Arch. Virol. 146 2369–2383
Gatehouse A M R, Powell K S, Peumans W J, Van Damme E J M and Gatehouse J A 1995 Insecticidal properties of plant lectins: their potential in plant protection; in Lectins: biomedical perspectives (eds) A Pusztai and S Bardocz (London, UK: Taylor and Francis) pp 35–57
Hester G, Kaku H, Goldstein I J and Wright C S 1995 Structure of mannose-specific snowdrop (Galanthus nivalis) lectin is representative of a new plant lectin family; Nat. Struct. Biol. 2 472–479
Hogervorst P A, Ferry N, Gatehouse A M, Wäckers F L and Romeis J 2006 Direct effects of snowdrop lectin (GNA) on larvae of three aphid predators and fate of GNA after ingestion; J. Insect Physiol. 52 614–624
Kaur A, Kamboj S S, Singh J, Singh R, Abrahams M, Kotwal G J and Saxena A K 2007 Purification of 3 monomeric monocot mannose-binding lectins and their evaluation for antipoxviral activity: potential applications in multiple viral diseases caused by enveloped viruses; Biochem. Cell Biol. 85 88–95
Keyaerts E, Vijgen L, Pannecouque C, Van Damme E J M, Peumans W J, Egberink H, Balzarini J and Van Ranst M 2007 Plant lectins are potent inhibitors of coronaviruses by interfering with two targets in the viral replication cycle; Antivir. Res. 75 179–187
Li K, Ooi V E C, Chuen C K Y, Lam A C, Ooi L S M, Zhang X B, Tsang K S, Chiu L C M, Chan K Y Y, Li C K, Fok T F, Yuen P M P and Ng P C 2008 The plant mannose-binding lectin NTL preserves cord blood haematopoietic stem/progenitor cells in long-term culture and enhances their ex vivo expansion; Br. J. Haematol. 140 90–98
Luo Y, Xu X, Liu J, Li J, Sun Y, Liu Z, Liu J, Van Damme E, Balzarini J and Bao J 2007 A novel mannose-binding tuber lectin from Typhonium divaricatum (L.) Decne (family Araceae) with antiviral activity against HSV-II and anti-proliferative effect on human cancer cell lines; J. Biochem. Mol. Biol. 40 358–367
Mosmann T J 1983 Rapid colorimetric assay for cellular growth and survival: application to proliferation and cytotoxic assays; J. Immunol. Methods 65 55
Ohizumi Y, Gaidamashvili M, Ohwada S, Matsuda K, Kominami J, Nakamura-Tsuruta S, Hirabayashi J, Naganuma T, Ogawa T and Muramoto K 2009 Mannose-binding lectin from yam (Dioscorea batatas) tubers with insecticidal properties against Helicoverpa armigera (Lepidoptera: Noctuidae); J. Agric. Food Chem. 57 2896–2902
Ooi L S M, Liu F, Ooi V E C, Ng T B and Fung M C 2002 Gene expression of immunomodulatory cytokines induced by Narcissus tazetta lectin in the mouse; Biochem. Cell Biol. 80 271–277
Ooi L S M, Ng T B, Geng Y and Ooi V E C 2000 Lectins from bulbs of the Chinese daffodil Narcissus tazetta (family Amaryllidaceae); Biochem. Cell Biol. 78 463–468
Ooi L S M, Sun S S M, Ng T B and Ooi V E C 2001 Molecular cloning and the cDNA-derived amino acid sequence of Narcissus tazetta isolectins; J. Protein Chem. 20 305–310
Ooi L S M, Sun S S M, Wang H and Ooi V E C 2004a New mannosebinding lectin isolated from the rhizome of Sarsaparilla Smilax glabra Roxb. (Liliaceae); J. Agric. Food Chem. 52 6091–6095
Ooi L S M, Sun S S M, and Ooi V E C 2004b Purification and characterization of a new antiviral protein from the leaves of Pandanus amaryllifolius (Pandanaceae); Int. J. Biochem. Cell Biol. 36 1440–1446
Ooi L S M, Wang H, Ng T B and Ooi V E C 1998 Isolation and characterization of a mannose-binding lectin from leaves of the Chinese daffodil Narcissus tazetta; Biochem. Cell Biol. 76 601–608
Serkedjieva J and Velcheva M 2003 In vitro anti-influenza virus activity of the pavine alkaloid (−)-thalimonine isolated from Thalictrum simplex L; Antivir. Chem. Chemother. 14 75–80
Smee D F, Huffman J H, Morrison A C, Barnard D L and Sidwell R W 2001 Cyclopentane neuraminidase inhibitors with potent in vitro anti-influenza virus activities; Antimicrob. Agents Chemother. 45 743–748
Smeets K, Van Damme E J M, Van Leuven F and Peumans W J 1997 Isolation, characterization and molecular cloning of a leafspecific lectin from ramsons (Allium ursinum L.); Plant Mol. Biol. 35 531–535
Tobita K, Sugiura A, Enomoto C and Furuyama M 1975 Plaque assay and primary isolation of influenza A viruses in an established line of canine kidney cells (MDCK) in the presence of trypsin; Med. Microbiol. Immunol. 162 9–14
Van Damme E J M, Kaku H, Perini F, Goldstein I J, Peters B, Yagi F, Decock B and Peumans W J 1991 Biosynthesis, primary structure and molecular cloning of snowdrop (Galanthus nivalis L.) lectin; Eur. J. Biochem. 202 23–30
Van Damme E J M, Smeets K and Peumans W J 1995 The mannose-binding monocot lectins and their genes; in A Pusztai and S Bardocz (eds) Lectins: biomedical perspectives (London, UK: Taylor and Francis) pp 59–80
Wu C F, An J, He X J, a Deng J, Hong Z X, Liu C, Lu H Z, Li Y J, Wang C J, Chen F and Bao J K 2004 Molecular cloning of a novel mannose-binding lectin gene from bulbs of Amaryllis vittata (Amaryllidaceae): Acta Bot. Sinica 46 1301–1306
Wu C F, Li J, An J, Chang L Q, Chen F and Bao J K 2006 Purification, biological activities, and molecular cloning of a novel mannose-binding lectin from bulbs of Zephyranthes candida Herb (Amaryllidaceae); J. Integr. Biol. 48 223–231
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ooi, L.S.M., Ho, WS., Ngai, K.L.K. et al. Narcissus tazetta lectin shows strong inhibitory effects against respiratory syncytial virus, influenza A (H1N1, H3N2, H5N1) and B viruses. J Biosci 35, 95–103 (2010). https://doi.org/10.1007/s12038-010-0012-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12038-010-0012-8