Abstract
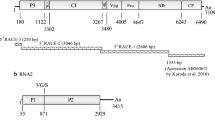
The complete nucleotide sequences of RNAs 1 and 2 of Rice stripe necrosis virus (RSNV) were determined and compared to the corresponding genomes of all sequenced, rod-shaped plant viruses. The genome organisation of RSNV RNA1 and RNA2 is nearly identical to that of Beet necrotic yellow vein virus (BNYVV) and Beet soil-borne mosaic virus (BSBMV), definitive species of the genus Benyvirus. As demonstrated for BNYVV and BSBMV, the RNA1 of RSNV also encodes a single ORF with putative replicase-associated motifs, which distinguishes benyviruses from all other viruses possessing rod-shaped particles. As described for BNYVV, RNSV RNA-2 also contains six ORFs: the capsid protein gene, the read-through protein gene, a triple gene block gene that codes for three different proteins, and a 17 kDa cysteine-rich protein. RNAs 3 and 4 (or 5 in the case of BNYVV), identified in natural infections of BNYVV and BSBMV, were not detected in any of the 44 RSNV cDNA clones obtained in this investigation. Nevertheless, phylogenetic and amino comparative acid sequence analyses demonstrated that RSNV is more closely related to BNYVV and BSBMV than to any other rod-shaped plant virus characterised to date.




Similar content being viewed by others
References
Bouzoubaa, S., Niesbach-Klösgen, U., Jupin, I., Guilley, H., Richards, K., & Jonard, G. (1991). Shortened forms of Beet necrotic yellow vein virus RNA-3 and -4: internal deletions and a subgenomic RNA. Journal of General Virology, 72, 259–266.
Fauquet, C. M., & Thouvenel, J. C. (1983). Association d’un nouveau virus en bâtonnet avec lamaladie de la nécrose a rayures du riz en Cote-d’Ivoire. Comptes Rendus de l’Academic des Sciences, 296, 575–578.
Fauquet, C. M., Mayo, M. A., Maniloff, J., Desselberger, U., & Ball, L. A. (eds). (2005). Virus taxonomy: Eigth report of the international committee on taxonomy of viruses. London: Elsevier.
Gilmer, D., Bouzoubaa, S., Hehn, A., Guilley, H., Richards, K., & Jonard, G. (1992). Efficient cell-to-cell movement of Beet necrotic yellow vein virus requires 3’proximal genes located on RNA 2. Virology, 189, 40–47.
Hall, M. C., & Matson, S. W. (1999). Helicase motifs: The engine that powers DNA unwinding. Molecular Microbiology, 34, 867–877.
Hehn, A., Fritsch, C., Richards, K. E., Guilley, H., & Jonard, G. (1997). Evidence for in vitro and in vivo autocatalytic processing of the primary translation product of Beet necrotic yellow vein virus RNA1 by a papain-like proteinase. Archives of Virology, 142, 1051–1058.
Koonin, E. V., & Dolja, V. V. (1993). Evolution and taxonomy of positive-strand RNA viruses: implications of comparative analysis of amino acid sequences. Critical Reviews in Biochemistry and Molecular Biology, 28, 375–430.
Lee, L., Telford, E. B., Batten, J. S., Scholthof, K.-B. G., & Rush, C. M. (2001). Complete nucleotide sequence and genome organization of Beet soil-borne mosaic virus, a proposed member of the genus Benyvirus. Archives of Virology, 146, 2443–2453.
Lemaire, O., Merdinoglou, D., Valentin, P., Putz, C., Ziegler-Graff, V., Guilley, H., et al. (1988). Effect of Beet necrotic yellow vein virus RNA composition on transmission by Polymyxa betae. Virology, 162, 232–235.
Louvel, D., & Bidaux, J. M. (1977). Observation de nouveaux symptômes pathologiques sur des variétés précoces de riz en Cote-d’Ivoire. Agronomie Tropicale, 32, 257–261.
Morales, F. J., Arroyave, J. A., Velasco, A. C., & Castaño, M. (1995). Caracterización parcial del ‘entorchamiento’ o ‘necrosis rayada’ del arroz en Colombia. Fitopatología Colombiana, 19, 48–54.
Morales, F. J. (2004). El ‘entorchamiento’ del arroz. Arrocero Moderno, 2004, 163–164.
Morales, F. J., Ward, E., Castaño, M., Arroyave, J. A., Lozano, I., & Adams, M. J. (1999). Emergence and partial characterization of Rice stripe necrosis virus and its fungus vector in South America. European Journal of Plant Pathology, 105, 643–650.
Poch, O., Sauvaget, I., Delarue, M., & Tordo, N. (1989). Identification of four conserved motifs among the RNA-dependent polymerase encoding elements. The EMBO Journal, 8, 3867–3874.
Putz, C. (2001). Composition and structure of Beet necrotic yellow vein virus. Journal of General Virology, 35, 397–401.
Quillet, L., Guilley, H., Jonard, G., & Richards, K. (1989). In vitro synthesis of biologically active Beet necrotic yellow vein virus RNA. Virology, 172, 293–301.
Rahim, M. D., Andika, I. B., ChenGui, H., Kondo, H., & Tamada, T. (2007). RNA4-encoded p31 of Beet necrotic yellow vein virus is involved in efficient vector transmission, symptom severity and silencing suppression in roots. Journal of General Virology, 88, 1611–1619.
Tamada, T., Shirako, Y., Abe, H., Saito, M., Kiguchi, T., & Harada, T. (1989). Production and pathogenicity of isolates of Beet necrotic yellow vein virus with different numbers of RNA components. Journal of General Virology, 70, 3399–3409.
Tamada, T., Schmitt, C., Saito, M., Guilley, H., Richards, K., & Jonard, G. (1996). High resolution analysis of the readthrough domain of Beet necrotic yellow vein virus readthrough protein: a KTER motif is important for efficient transmission of the virus by Polymyxa betae. Journal of General Virology, 77, 1359–1367.
Torrance, L., & Mayo, M. (1997). Proposed re-classification of furoviruses. Archives of Virology, 142, 435–439.
Van Regenmortel, M. H. V., Fauquet, C. M., Bishop, D. H. L., Carstens, E. B., Estes, M. K., Lemon, S. M., et al. (eds). (2000). Virus taxonomy: Seventh report of the international committee on taxonomy of viruses. New York: Academic.
Acknowledgements
The authors thank Camilo Oliveros for assistance with the graphic materials presented here.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Lozano, I., Morales, F. Molecular characterisation of Rice stripe necrosis virus as a new species of the genus Benyvirus . Eur J Plant Pathol 124, 673–680 (2009). https://doi.org/10.1007/s10658-009-9453-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10658-009-9453-z