Abstract
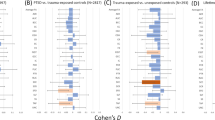
Gulf War illness (GWI) is a chronic disease characterized by the involvement of several organs, including the brain (Christova et al., Exp Brain Res doi:10.1007/s00221-017-5010-8, 2017). In a previous study (Georgopoulos et al., J Neural Eng 4:349–355, 2015), we identified six protective alleles from Class II human leukocyte antigen (HLA) genes, and more recently, we investigated the brain correlates of this protection (James et al., EBioMedicine 13:72–79, 2016). Those and other studies (Israeli, Lupus, 21:190–194, 2012) suggested an involvement of the immune system in GWI. In a recent study (Engdahl et al., EBioMedicine doi:10.1016/j.ebiom.2016.08.030, 2016), we showed that the brain pattern of synchronous neural interactions (SNI; Georgopoulos et al., J Neural Eng 4:349–355, 2007) in GWI is distinctly different from that in healthy controls. Here we focused on the SNI itself, as a basic measure of neural communication (irrespective of specific connections) and compared it between GWI and seven other diseases that cover a broad spectrum of etiology and pathophysiology. Specifically, we sought to determine which, if any, of those diseases might resemble GWI SNI, overall and within the HLA protective domain, and thus gain further knowledge regarding the nature of GWI brain abnormality. We studied a total of 962 participants from a healthy control population (N = 583) and eight different diseases, including GWI (N = 40), schizophrenia (SZ; N = 21), Alzheimer’s disease (AD; N = 66), posttraumatic stress disorder (PTSD; N = 159), major depressive disorder (MDD; N = 10), relapsing–remitting multiple sclerosis (RRMS; N = 43), Sjögren’s syndrome (SS; N = 32), and rheumatoid arthritis (RA; N = 8). They all underwent a resting-state magnetoencephalographic (MEG) scan to calculate SNIs. Data were analyzed using analysis of covariance (ANCOVA) with disease as fixed factor, and sex and age as covariates. We found that GWI SNIs differed significantly from control SZ, AD, PTSD and MDD but not from RRMS, SS and RA. In addition, we compared GWI to RRMS, SS and RA with respect to SNIs of MEG sensor pairs that were related to the HLA alleles protective for GWI (James et al., EBioMedicine 13:72–79, 2016). We found that GWI SNIs did not differ significantly from any of these three diseases but they did so from control SZ, AD, PTSD and MDD. These findings indicate that (a) GWI brain synchronicity does not differ significantly from that of known immune-related diseases (RRMS, SS, RA), and (b) that this SNI similarity is present within the HLA-related SNIs. In contrast, GWI SNIs differed significantly from those of the other diseases. We conclude that altered brain communication in GWI likely reflects immune-related processes, as postulated previously (James et al., EBioMedicine 13:72–79, 2016). By extension, these findings also indicate that functional brain abnormalities in RRMS, SS and RA might be, in part, due to lack of protective HLA alleles as documented for GWI (Georgopoulos et al., EBioMedicine 3:79–85, 2015).




Similar content being viewed by others
References
Ainala H, Loukkola J, Peltola J et al (2001) The prevalence of neuropsychiatric syndromes in systemic lupus erythematosus. Neurology 57:496–500
Alexander E, Provost TT (1987) Sjögren’s syndrome: association of cutaneous vasculitis with central nervous system disease. Arch Dermatol 123:801–810
Amato MP, Zipoli V, Portaccio E (2006) Multiple sclerosis-related cognitive changes: a review of cross-sectional and longitudinal studies. J Neurol Sci 245:41–46
American Psychiatric Association (2000) Diagnostic and statistical manual of mental disorders, 4th edn. APA, Washington, DC
Anders SL, Peterson CK, James LM et al (2015) Neural communication in posttraumatic growth. Exp Brain Res 233:2013–2020
Antonchak MA, Saoudian M, Khan AR et al (2011) Cognitive dysfunction in patients with systemic lupus erythematosus: a controlled study. J Rheumatol 38:1020–1025
Blake D, Weathers F, Nagy LM et al (1995) Clinician-Administered PTSD Scale. National Center for PTSD, Boston
Box GEP, Jenkins GM (1976) Time series analysis: forecasting and control. Holden-Day, San Francisco
Carbotte RM, Denburg SD, Denburg JA (1986) Prevalence of cognitive impairment in systemic lupus erythematosus. J Nervous Mental Dis 174:357–364
Chiaravalloti ND, DeLuca J (2008) Cognitive impairment in multiple sclerosis. Lancet Neurol 7:1139–1151
Christova P, James LM, Engdahl BE, Lewis SM, Georgopoulos AP (2015) Diagnosis of posttraumatic stress disorder (PTSD) based on correlations of prewhitened fMRI data: outcomes and areas involved. Exp Brain Res 233:2695–2705
Christova P, James LM, Engdahl BE et al (2017) Subcortical brain atrophy in Gulf War Illness. Exp Brain Res. doi:10.1007/s00221-017-5010-8 (Epub ahead of print)
de Melo LF, Da-Silva SL (2012) Neuropsychological assessment of cognitive disorders in patients with fibromyalgia, rheumatoid arthritis, and systemic lupus erythematosus. Brazil J Rheumatol 52:175–188
Denney DR, Sworowski LA, Lynch SG (2005) Cognitive impairment in three subtypes of multiple sclerosis. Arch Clin Neuropsychol 20:967–981
Engdahl BE, Leuthold A, Tan HR et al (2010) Post-traumatic stress disorder: a right temporal lobe syndrome? J Neural Eng 7:066005
Engdahl BE, James LM, Miller RD et al (2016) A magnetoencephalographic (MEG) study of Gulf War Illness (GWI). EBioMedicine. doi:10.1016/j.ebiom.2016.08.030
First MB, Spitzer RL, Gibbon M et al (2002) Structural clinical interview for DSM-IV-TR axis I disorders, research version, non-patient edition (SCID-I/NP). Biometrics Research, New York State Psychiatric Institute, New York
Fisher RA (1958) Statistical methods for research workers, 13th edn. Oliver and Boyd, Edinburgh
Fukuda K, Nisenbaum R, Stewart G et al (1998) Chronic multisymptom illness affecting Air Force veterans of the Gulf War. JAMA 280:981–988
Furukawa H, Oka S, Tsuchiya N et al (2017) The role of common protective alleles HLA-DRB1*13 among systemic autoimmune diseases. Genes Immun 18:1–7
Georgopoulos AP, Karageorgiou E, Leuthold A et al (2007) Synchronous neural interactions assessed by magnetoencephalography: a functional biomarker for brain disorders. J Neural Eng 4:349–355
Georgopoulos AP, Tan HM, Lewis SM et al (2010) The synchronous neural interactions test as a functional neuromarker for post-traumatic stress disorder (PTSD): a robust classification method based on the bootstrap. J Neural Engin 7:016011
Georgopoulos AP, James LM, Mahan MY et al (2015) Reduced Human Leukocyte Antigen (HLA) protection in Gulf War Illness (GWI). EBioMedicine 3:79–85
Ginsburg KS, Wright EA, Larson MG et al (1992) A controlled study of the prevalence of cognitive dysfunction in randomly selected patients with systemic lupus erythematosus. Arthritis Rheum 35:776–782
Hamed SA, Selim ZI, Elattar AM et al (2012) Assessment of biocorrelates for brain involvement in female patients with rheumatoid arthritis. Clin Rheumatol 31:123–132
Hanly JG, Fisk JD, Eastwood B (1994) Brain reactive autoantibodies and cognitive impairment in systemic lupus erythematosus. Lupus 3:193–199
Hanly JG, Fisk JD, McCurdy G et al (2005) Neuropsychiatric syndromes in patients with systemic lupus erythematosus and rheumatoid arthritis. J Rheumatol 32:1459–1466
Hay EM, Black D, Huddy A et al (1992) Psychiatric disorder and cognitive impairment in systemic lupus erythematosus. Arthritis Rheum 35:411–416
Heaton RK, Nelson LM, Thompson DS et al (1985) Neuropsychological findings in relapsing-remitting and chronic-progressive multiple sclerosis. J Consul Clin Psychol 53:103
Hotopf M, David A, Hull L et al (2000) Role of vaccinations as risk factors for ill health in veterans of the Gulf war: cross-sectional study. BMJ 320:1363–1367
Israeli E (2012) Gulf War Syndrome as a part of the autoimmune (autoinflammatory) syndrome induced by adjuvant (ASIA). Lupus 21:190–194
James LM, Engdahl BE, Leuthold AC et al (2012) Neural network modulation by trauma as a marker of resilience: differences between veterans with posttraumatic stress disorder and resilient controls. JAMA Psychiatry 70:410–418
James LM, Belitskaya-Levy I, Lu Y et al (2014) Development and application of a diagnostic algorithm for posttraumatic stress disorder. Psychiatry Res 231:1–7
James LM, Engdahl BE, Leuthold AC, Krueger RF, Georgopoulos AP (2015) Pathological personality traits modulate neural interactions. Exp Brain Res 233:3543–3552
James LM, Engdahl BE, Leuthold AC, Georgopoulos AP (2016) Brain correlates of human leukocyte antigen (HLA) protection in Gulf War Illness (GWI). EBioMedicine 13:72–79
Kozora E, Laudenslager M, Lemieux A, West SG (2001) Inflammatory and hormonal measures predict neuropsychological functioning in systemic lupus erythematosus and rheumatoid arthritis patients. J Int Neuropsychol Soc 7:745–754
Lafitte C, Amoura Z, Cacoub P et al (2001) Neurological complications of primary Sjögren’s syndrome. J Neurol 248:577–584
Langheim FPJ, Leuthold AC, Georgopoulos AP (2006) Synchronous dynamic brain networks revealed by magnetoencephalography. Proc Natl Acad Sci USA 103:455–459
Leuthold AC, Mahan MY, Stanwyck JJ, Georgopoulos A, Georgopoulos AP (2013) The number of cysteine residues per mole in apolipoprotein E affects systematically synchronous neural interactions in women’s healthy brains. Exp Brain Res 226:525–536
Lewis SM, Vydrová RR, Leuthold AC, Georgopoulos AP (2016) Cortical miscommunication after prenatal exposure to alcohol. Exp Brain Res 234:3347–3353
Mahan MY, Chorn CR, Georgopoulos AP (2015) White Noise Test: detecting autocorrelation and nonstationarities in long time series after ARIMA modeling. Proc 14th Python In Science Conference (Scipy 2015), Austin, TX
Martinez S, Caceres C, Mataro M et al (2010) Is there progressive cognitive dysfunction in Sjögren Syndrome? A preliminary study. Acta Neurol Scand 122:182–188
McKhann G, Drachman D, Folstein M et al (1984) Clinical diagnosis of Alzheimer’s disease Report of the NINCDS-ADRDA Work Group under the auspices of Department of Health and Human Services Task Force on Alzheimer’s Disease. Neurology 34:939–944
Meuer SC, Hussey RE, Hodgdon JC et al (1982) Surface structures involved in target recognition by human cytotoxic T lymphocytes. Science 218:471–473
Moss JI (2013) Gulf War illnesses are autoimmune illnesses caused by increased activity of the p38/MAPK pathway in CD4+ immune system cells, which was caused by nerve agent prophylaxis and adrenergic load. Med Hypotheses 81:1002–1003
Parkitny L, Middleton S, Baker K, Younger J (2015) Evidence for abnormal cytokine expression in Gulf War Illness: a preliminary analysis of daily immune monitoring data. BMC Immunol. doi:10.1186/s12865-015-0122-z
Polman CH, Reingold SC, Edan G et al (2005) Diagnostic criteria for multiple sclerosis: 2005 revisions to the “McDonald Criteria”. Annals Neuro 58:840–846
Priestley MB (1981) Spectral analysis of time series. Academic, San Diego
Rao SM, Leo GJ, Bernardin L, Unverzagt F (1991) Cognitive dysfunction in multiple sclerosis. I. Frequency, patterns, and prediction. Neurology 41:685–691
Segal BM, Pogatchnik B, Holker E et al (2012) Primary Sjogren’s syndrome: cognitive symptoms, mood, and cognitive performance. Acta Neurol Scand 125:272–278
Segal BM, Rhodus N, Sivils KLM, Solid CA (2014) Validation of the brief cognitive symptoms index in sjögren syndrome. J Rheumatol 41:2027–2033
Shin SY, Katz P, Wallhagen M, Julian L (2012) Cognitive impairment in persons with rheumatoid arthritis. Arthritis Care Res 64:1144–1150
Shin SY, Julian L, Katz P (2013) The relationship between cognitive function and physical function in rheumatoid arthritis. J Rheumatol 40:236–243
Singer W (1999) Neuronal synchrony: a versatile code for the definition of relations? Neuron 24:49–65
Skowera A, Hotopf M, Sawicka E et al (2004) Cellular immune activation in Gulf War veterans. J Clin Immunol 24:66–73
Steele L (2000) Prevalence and patterns of Gulf War illness in Kansas veterans: association of symptoms with characteristics of person, place, and time of military service. Am J Epidemiol 152:992–1002
Uhlhaas PJ, Singer W (2006) Neural synchrony in brain disorders: relevance for cognitive dysfunctions and pathophysiology. Neuron 52:155–168
Vitali C, Bombardieri S, Jonsson R (2002) Classification criteria for Sjögren’s syndrome: a revised version of the European criteria proposed by the American-European Consensus Group. Annals Rheum Dis 61:554–558
Whistler T, Fletcher MA, Lonergan W et al (2009) Impaired immune function in Gulf War illness. BMC Med Genom 2:1. doi:10.1186/1755-8794-2-12
White RF, Steele L, O’Callaghan JP et al (2016) Recent research on Gulf War illness and other health problems in veterans of the 1991 Gulf War: effects of toxicant exposures during deployment. Cortex 74:449–475
Acknowledgements
This work was partially supported by a service directed grant from the United States Department of Veterans Affairs, a grant for the United States Department of Defense (Award Number W81XWH-15-1-0520), and the American Legion Brain Sciences Chair. The contents do not represent the views of the U.S. Department of Veterans Affairs or the United States Government.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors do not report any financial disclosures or conflicts of interest.
Rights and permissions
About this article
Cite this article
Georgopoulos, A.P., James, L.M., Carpenter, A.F. et al. Gulf War illness (GWI) as a neuroimmune disease. Exp Brain Res 235, 3217–3225 (2017). https://doi.org/10.1007/s00221-017-5050-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00221-017-5050-0