Abstract
Preclinical cell-based toxicity screening is an essential stage in the drug development process. Current technology is based on immortalized cell lines, rodent cells and primary human hepatocytes, all of which suffer from shortcomings. Cell lines and rodent cells have limited relevance to human physiology, while primary human cells remain a scarce and variable resource. These systems are inadequate, as evidenced by the high levels of compound attrition in the clinical trial and postmarketing stages of development, producing safety risks and high financial costs. Hepatotoxicity and drug-induced liver injury account for a substantial proportion of compound failures, highlighting the need for accurate and predictive liver toxicity models. Pluripotent stem cell-derived hepatocyte-like cells offer a means of creating physiologically relevant drug screening assays that could serve as an additional method of detecting toxicity in the lead optimization phase of drug development. The scalability and definition of pluripotent cell culture systems are constantly improving, bringing a potentially inexhaustible cell resource closer to industrial translation. Meanwhile, increased understanding of pluripotency, differentiation and reprogramming, combined with optimization of tissue culture environments, will allow the ongoing issues of hepatocyte lifespan and immature function to be addressed. In future, extensive validation of stem cell-derived hepatocyte-like cells against existing drug screening methods will be required if they are to be established as a standard tool for investigating drug toxicity.
Similar content being viewed by others
References
Gómez-Lechón MJ, Lahoz A, Gombau L, et al. In vitro evaluation of potential hepatotoxicity induced by drugs. Curr Pharm Des 2010; 16 (17): 1963–77 2. Orloff J, Douglas F, Pinheiro J, et al. The future of drug development: advancing clinical trial design. Nat Rev Drug Discov 2010; 8 (12): 949–57
Orloff J, Douglas F, Pinheiro J, et al. The future of drug development: advancing clinical trial design. Nat Rev Drug Discov 2010; 8 (12): 949–57
Lee WM. Acute liver failure in the United States. Semin Liver Dis 2003; 23 (3): 217–26
Wong N, Lai P, Pang E, et al. A comprehensive karyotypic study on human hepatocellular carcinoma by spectral karyotyping. Hepatology 2000; 32 (5): 1060–8
Lake BG. Species differences in the hepatic effects of inducers of CYP2B and CYP4A subfamily forms: relationship to rodent liver tumour formation. Xenobiotica 2009; 39: 582–96, No. 8
Hewitt NJ, Lechon MJ, Houston JB, et al. Primary hepatocytes: current understanding of the regulation of metabolic enzymes and transporter proteins, and pharmaceutical practice for the use of hepatocytes in metabolism, enzyme induction, transporter, clearance, and hepatotoxicity studies. Drug Metab Rev 2007; 39 (1): 159–234
Miranda J, Leite S, Muller-Vieira U, et al. Towards an extended functional hepatocyte in vitro culture. Tissue Eng Part C: Methods 2009; 15 (2): 157–67
Reubinoff BE, Pera MF, Fong CY, et al. Embryonic stem cell lines from human blastocysts: somatic differentiation in vitro. Nat Biotechnol 2000; 18: 399–404
Thomson JA, Itskovitz-Eldor J, Shapiro SS, et al. Embryonic stem cell lines derived from human blastocysts. Science 1998; 282: 1145–7
Takahashi K, Tanabe K, Ohnuki M, et al. Induction of pluripotent stem cells from adult human fibroblasts by defined factors. Cell 2007; 131 (5): 861–72
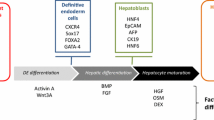
Hay DC, Fletcher J, Payne C, et al. Highly efficient differentiation of hESCs to functional hepatic endoderm requires ActivinA and Wnt3a signaling. Proc Natl Acad Sci U S A 2008; 105 (34): 12301–6
Hay DC, Pernagallo S, Diaz-Mochon JJ, et al. Unbiased screening of polymer libraries to define novel substrates for functional hepatocytes with inducible drug metabolism. Stem Cell Res 2011; 6 (2): 92–102
Medine CN, Lucendo-Villarin B, Zhou W, et al. Robust generation of metabolically active hepatocytes from pluripotent stem cells. J Vis Exp 2011 (56), e2969 [online]. Available from URL: http://www.jove.com/video/2969/robustgeneration-of-hepatocyte-like-cells-from-human-embryonic-stem-cell-populations [Accessed 2012 Jan 12]
Kola I, Landis J. Can the pharmaceutical industry reduce attrition rates? Nat Rev Drug Discov 2004; 3 (8): 711–5
Goldring CE, Kitteringham NR, Jenkins R, et al. Development of a transactivator in hepatoma cells that allows expression of phase I, phase II, and chemical defense genes. Am J Physiol Cell Physiol 2006; 290 (1): C104–15
Bissell DM, Gores GJ, Laskin DL, et al. Drug-induced liver injury: mechanisms and test systems. Hepatology 2001; 33 (4): 1009–13
Usui T, Mise M, Hashizume T, et al. Evaluation of the potential for drug-induced liver injury based on in vitro covalent binding to human liver proteins. Drug Metab Dispos 2009; 37 (12): 2383–92
Cheng N, Wauthier E, Reid L. Mature human hepatocytes from ex vivo differentiation of alginate-encapsulated hepatoblasts. Tissue Eng Part A 2008; 14 (1): 1–7
Fox I, Strom S. To be or not to be: generation of hepatocytes from cells outside the liver. Gastroenterology 2008; 134 (3): 878–81
Hart SN, Li Y, Nakamoto K, et al. A comparison of whole genome gene expression profiles of HepaRG cells and HepG2 cells to primary human hepatocytes and human liver tissues. Drug Metab Dispos 2010; 38 (6): 988–94
Zanelli U, Caradonna NP, Hallifax D, et al. Comparison of cryopreserved HepaRG cells with cryopreserved human hepatocytes for prediction of clearance for 26 drugs. Drug Metab Dispos 2012; 40 (1): 104–10
Weber A, Touboul T, Mainot S, et al. Human foetal hepatocytes: isolation, characterization, and transplantation. Methods Mol Biol 2010; 640: 41–55
Gale EA. Lessons from the glitazones: a story of drug development. Lancet 2001 9; 357 (9271): 1870–5
Park IH, Arora N, Huo H, et al. Disease-specific induced pluripotent stem cells. Cell 2008; 134 (5): 877–86
Xie HG, Kim RB, Wood AJJ, et al. Molecular basis of ethnic differences in drug disposition and response. Annu Rev Pharmacol Toxicol 2001; 41: 815–50
EUR-Lex. Judgment of the court (grand chamber) of 18 October 2011. Oliver Brüstle v Greenpeace eV [online]. Available from URL: http://eur-lex.europa.eu/LexUriServ/LexUriServ.do?uri=CELEX:62010J0034:EN:NOT [Accessed 2012 Jan 12]
Itskovitz-Eldor J, Schuldiner M, Karsenti D, et al. Differentiation of human embryonic stem cells into embryoid bodies compromising the three embryonic germ layers. Mol Med 2000; 6 (2): 88–95
Basma H, Soto-Gutiérrez A, Yannam G, et al. Differentiation and transplantation of human embryonic stem cell-derived hepatocytes. Gastroenterology 2009; 136 (3): 990–9
Baharvand H, Hashemi SM, Kazemi Ashtiani S, et al. Differentiation of human embryonic stem cells into hepatocytes in 2D and 3D culture systems in vitro. Int J Dev Biol 2006; 50 (7): 645–52
Imamura T, Cui L, Teng R, et al. Embryonic stem cell-derived embryoid bodies in three-dimensional culture system form hepatocyte-like cells in vitro and in vivo. Tissue Eng 2004; 10 (11–12): 1716–24
Hannoun Z, Fletcher J, Greenhough S, et al. The comparison between conditioned media and serum free media in human embryonic stem cell culture and differentiation. Cell Reprog 2010; 12 (2): 133–40
Sullivan GJ, Hay DC, Park IH, et al. Generation of functional human hepatic endoderm from human induced pluripotent stem cells. Hepatology 2010; 51 (1): 329–35
Agarwal S, Holton K, Lanza R. Efficient differentiation of functional hepatocytes from human embryonic stem cells. Stem Cells 2008; 26 (5): 1117–27
Duan Y, Catana A, Meng Y, et al. Differentiation and enrichment of hepatocyte- like cells from human embryonic stem cells in vitro and in vivo. Stem Cells 2007; 25 (12): 3058–68
Brolén G, Sivertsson L, Bjö rquist P, et al. Hepatocyte-like cells derived from human embryonic stem cells specifically via definitive endoderm and a progenitor stage. J Biotechnol 2010 1; 145 (3): 284–94
Touboul T, Hannan NR, Corbineau S, et al. Generation of functional hepatocytes from human embryonic stem cells under chemically defined conditions that recapitulate liver development. Hepatology 2010; 51 (5): 1754–65
Si-Tayeb K, Noto FK, Nagaoka M, et al. Highly efficient generation of human hepatocyte-like cells from induced pluripotent stem cells. Hepatology 2010; 51 (1): 297–305
Rashid ST, Corbineau S, Hannan N, et al. Modeling inherited metabolic disorders of the liver using human induced pluripotent stem cells. J Clin Invest 2010;120 (9): 3127–36
Asgari S, Pournasr B, Salekdeh GH, et al. Induced pluripotent stem cells: a new era for hepatology. J Hepatol 2010; 53 (4): 738–51
Huang P, He Z, Ji S, et al. Induction of functional hepatocyte-like cells from mouse fibroblasts by defined factors. Nature 2011 11; 475 (7356): 386–9
Sekiya S, Suzuki A. Direct conversion of mouse fibroblasts to hepatocyte-like cells by defined factors. Nature 2011 29; 475 (7356): 390–3
Guttman M, Donaghey J, Carey BW, et al. lincRNAs act in the circuitry controlling pluripotency and differentiation. Nature 2011 28; 477 (7364): 295–300
Discher DE, Janmey P, Wang Y. Tissue cells feel and respond to the stiffness of their substrate. Science 2005; 310 (5751): 1139–43
Engler AJ, Sen S, Sweeney HL, et al. Matrix elasticity directs stem cell lineage specification. Cell 2006 25; 126 (4): 677–89
Dalby MJ, Gadegaard N, Curtis AS, et al. Nanotopographical control of human osteoprogenitor differentiation. Curr Stem Cell Res Ther 2007; 2 (2): 129–38
Machide M, Hashigasako A, Matsumoto K, et al. Contact inhibition of hepatocyte growth regulated by functional association of the c-Met/hepatocyte growth factor receptor and LAR protein-tyrosine phosphatise. J Biol Chem 2006 31; 281 (13): 8765–72
Iredale JP, Arthur MJ. Hepatocyte-matrix interactions. Gut 1994; 35 (6): 729–32
Zweigerdt R, Olmer R, Singh H, et al. Scalable expansion of human pluripotent stem cells in suspension culture. Nat Protoc 2011; 6 (5): 689–700
Olmer R, Haase A, Merkert S, et al. Long term expansion of undifferentiated human iPS and ES cells in suspension culture using a defined medium. Stem Cell Res 2010; 5 (1): 51–64
Acknowledgements
Mr Sebastian Greenhough was supported by a Knowledge Transfer Partnership award and Roslin Cellab Ltd. Mr Greenhough has no other conflicts of interest to declare. Dr David Hay was supported by a Research Councils UK fellowship. Dr David Hay is a Director, Chief Scientific Officer and Shareholder in FibromEd Limited.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Greenhough, S., Hay, D.C. Stem Cell-Based Toxicity Screening. Pharm Med 26, 85–89 (2012). https://doi.org/10.1007/BF03256896
Published:
Issue Date:
DOI: https://doi.org/10.1007/BF03256896